Pyruvate Dehydrogenase & Krebs
Cycle
Pathway
localization:
Glycolysis enzymes are located in the cytosol of
cells. Pyruvate enters the mitochondrion to be metabolized further.
|
Mitochondrial compartments:
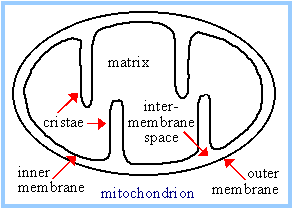
The mitochondrial matrix contains
Pyruvate Dehydrogenase and enzymes of Krebs Cycle, plus other pathways such
as fatty acid oxidation. The mitochondrial outer membrane contains large
VDAC channels, similar to bacterial
porin
channels, making the outer membrane leaky to ions and small molecules.
The inner membrane is the major
permeability barrier of the mitochondrion. It contains various transport
catalysts, including a carrier protein that allows pyruvate to enter the
matrix. It is highly convoluted, with infoldings called cristae. Embedded in
the inner membrane are constituents of the
respiratory chain
and
ATP Synthase.
|
|
|
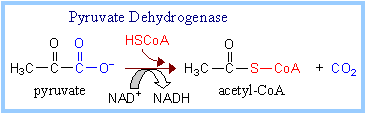
Pyruvate Dehydrogenase
catalyzes oxidative decarboxylation of pyruvate, to form acetyl-CoA. The
overall reaction is shown at right.
|
|
|
Pyruvate Dehydrogenase is a
large complex
containing many copies of each of three enzymes, E1, E2,
and E3. The structure of the complex is as follows:
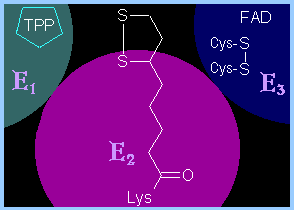
The inner core of the mammalian Pyruvate
Dehydrogenase complex is an icosahedral structure consisting of 60 copies of
E2.
At the periphery of the complex are:
- 30 copies of
E1 (itself a
tetramer with subunits a2b2)
and
- 12 copies of
E3 (a
homodimer), plus 12 copies of an E3 binding protein that links
E3 to E2.
Prosthetic groups are listed below, a cartoon
showing 3 subunits is at right, and a diagram summarizing the reactions
catalyzed is on p. 770. |
|
|
Enzyme |
Abbreviated |
Prosthetic Group |
|
Pyruvate Dehydrogenase |
E1 |
Thiamine pyrophosphate (TPP) |
|
Dihydrolipoyl
Transacetylase |
E2 |
Lipoamide |
|
Dihydrolipoyl Dehydrogenase |
E3 |
FAD |
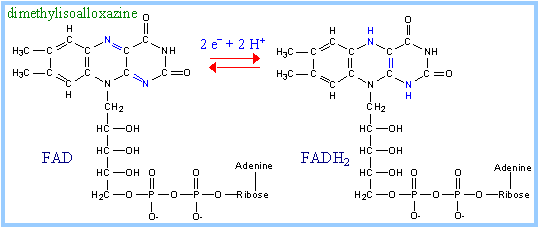
FAD (Flavin
Adenine Dinucleotide)
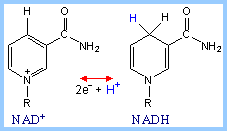
is a derivative of the B-vitamin riboflavin (dimethylisoalloxazine-ribitol). The
flavin ring system undergoes oxidation/reduction as shown below. Whereas NAD+
is a coenzyme that reversibly binds to enzymes, FAD is a
prosthetic group, that is
permanently part of the complex.
|
FAD accepts and donates 2
electrons with 2 protons (2 H):
FAD + 2 e- + 2 H+
¨ FADH2 |
|
Thiamine pyrophosphate (TPP) is a derivative of
thiamine (vitamin B1). Nutritional deficiency of thiamine leads to
the disease beriberi.
Beriberi affects especially the brain, because TPP is required for carbohydrate
metabolism, and the brain depends on glucose metabolism for energy.
|
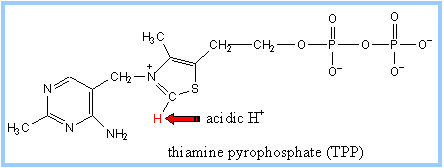 
A proton readily dissociates from the
C that is between
N and
S in the thiazole ring of
TPP. The resulting carbanion (ylid) can attack the electron-deficient keto
carbon of pyruvate. |
|
|
Lipoamide includes a dithiol that undergoes oxidation and reduction.
The carboxyl group at the end of lipoic
acid's hydrocarbon chain forms an amide bond to the side-chain amino group
of a lysine residue of E2.
A long flexible arm, including hydrocarbon
chains of lipoate and the lysine R-group, links the dithiol of each
lipoamide to one of two lipoate-binding domains of each
E2. Lipoate-binding
domains are themselves part of a flexible strand of E2 that
extends out from the core of the complex.
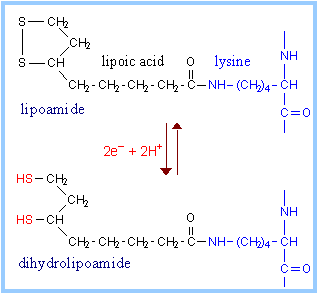
The long flexible attachment allows lipoamide
functional groups to swing back and forth between E2 active sites in the
core of the complex and active sites of E1 & E3 in the outer shell of the
complex.
The E3 binding protein (that binds E3 to E2)
also has attached lipoamide that can exchange reducing equivalents with
lipoamide on E2.
For diagrams showing the approximate
arrangement of functional domains, based on structural studies of Pyruvate
Dehydrogenase and a related enzyme see:
- a
website
of the laboratory of Wim Hol.
- an
article
by Milne et al. (Fig. 5, requires a
subscription to J. Biol. Chem.).
|
|
|
Organic arsenicals are
potent inhibitors of lipoamide-containing enzymes such as Pyruvate
Dehydrogenase. These highly toxic compounds react with "vicinal" dithiols
such as the functional group of lipoamide as shown at right.
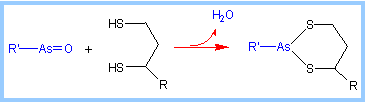
|
|
|
In the overall reaction,
the acetic acid generated is transferred to
coenzyme A.
The final electron acceptor is NAD+.
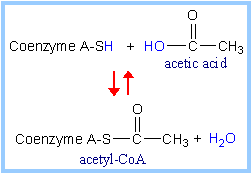 
|
|
|
|
The reaction proceeds as follows: |
- The keto carbon of
pyruvate reacts with the
carbanion of TPP on
E1 to
yield an addition compound. The electron-pulling positively charged nitrogen
of the thiazole ring promotes loss of CO2. What remains is
hydroxyethyl-TPP.
- The hydroxyethyl carbanion
on TPP of E1
reacts with the disulfide of
lipoamide on E2.
What was the keto carbon of pyruvate is oxidized to a carboxylic acid, as the
disulfide of lipoamide is reduced to a dithiol. The
acetate formed by oxidation
of the hydroxyethyl moiety is linked to one of the thiols of the reduced
lipoamide as a thioester
(~).
- The acetate is transferred
from the thiol of lipoamide to the thiol of
coenzyme A, yielding
acetyl CoA.
- The reduced lipoamide swings
over to the E3
active site. Dihydrolipoamide is reoxidized to the disulfide, as 2 e-
+ 2 H+ are transferred to a disulfide on E3 (disulfide
interchange).
- The dithiol on E3
is reoxidized as 2 e- + 2 H+ are transferred to
FAD. The resulting FADH2
is reoxidized by electron transfer to
NAD+, to yield
NADH + H+.
|
Acetyl CoA, a product of the
Pyruvate Dehydrogenase reaction, is a central compound in metabolism. The
"high energy" thioester linkage makes it an excellent donor of the acetate
moiety.
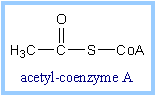
|
|
|
For example, acetyl CoA
functions as:
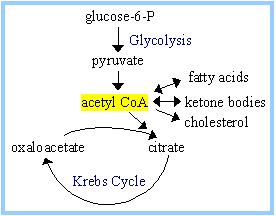
Regulation of Pyruvate Dehydrogenase
complex : |
|
Product inhibition
by NADH and acetyl CoA: NADH competes with NAD+ for
binding to E3. Acetyl CoA competes with Coenzyme A for binding to E2.
Regulation by phosphorylation/dephosphorylation
of E1: Specific regulatory Kinases and Phosphatases are associated
with the Pyruvate Dehydrogenase complex within the mitochondrial matrix.
- Pyruvate Dehydrogenase
Kinases catalyze phosphorylation of serine residues of E1,
inhibiting the complex.
- Pyruvate Dehydrogenase
Phosphatases reverse this inhibition.
Pyruvate Dehydrogenase Kinases
are activated by NADH and acetyl-CoA, providing another way the two major
products of the Pyruvate Dehydrogenase reaction inhibit the complex. Pyruvate
Dehydrogenase Kinase activation involves interaction with E2 subunits
to sense changes in oxidation state and acetylation of lipoamide caused by NADH
and acetyl-CoA.
During starvation, Pyruvate Dehydrogenase Kinase
increases in amount in most tissues, including skeletal muscle, via increased
gene transcription. Under the same conditions, the amount of Pyruvate
Dehydrogenase Phosphatase decreases. The resulting inhibition of Pyruvate
Dehydrogenase prevents muscle and other tissues from catabolizing glucose and
gluconeogenesis precursors. Metabolism shifts toward fat utilization, while
muscle protein breakdown to supply gluconeogenesis precursors is minimized, and
available glucose is spared for use by the brain.
A Ca++-sensitive isoform of the
phosphatase that
removes phosphate residues from E1 is expressed in muscle cells. The
increased cytosolic Ca++ that occurs during activation of muscle
contraction can lead to Ca++ uptake by mitochondria. The higher Ca++
stimulates the phosphatase, and dephosphorylation activates Pyruvate
Dehydrogenase. Thus mitochondrial metabolism may be stimulated during exercise.
|
Lecture
notes relating to Krebs Cycle are not provided in the usual format, because
lectures will be presented by students. Some questions on Krebs Cycle are
included in the self-study quiz for this class.
Select the interactive tutorial at right for
information about the Krebs Citric Acid Cycle. Within the tutorial, drag the
cursor over each enzyme name for information about that reaction.
Note that FADH2, listed as a
product of succinate oxidation, is reoxidized to FAD as redox carriers
within the Succinate Dehydrogenase complex pass electrons to coenzyme Q of
the
respiratory chain.
Thus it would be more appropriate to list coenzyme QH2 as a
product of the Succinate Dehydrogenase reaction. The initial acceptor, FAD,
is included in the diagram for consistency with most textbooks. |
|
KREBS CYCLE OR TRICARBOXYLIC
ACID CYCLE OR TCA OR CITRIC ACID CYCLE:
This referred as
cycle because oxaloacetate regenerated at the end of each cycle and it is also
one of the initial substrate enter into the cycle. This cycle occurs within the
mitochondrial matrix. This is a series of biochemical reactions aerobic
organisms use to release chemical energy stored in acetyl CoA. Acetyl CoA is a
product of catabolic reactions in carbohydrate, lipid and amino acid
metabolism. In this cycle, the carbon atoms derived from the acetyl group of
acetyl coA are oxidized to CO2. The high energy electrons removed from citric
acid cycle intermediates are transferred to NAD+ and FAD to form the reduced
coenzymes NADH and FADH2. In one of the step, the high energy molecule
guanosine triphosphate (GTP) is produced during a substrate level
phosphorylation. The overall reaction for TCA cycle is as follows:
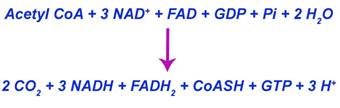
Krebs cycle occurs
in eight steps. The detailed reactions of cycle are shown in the figure.
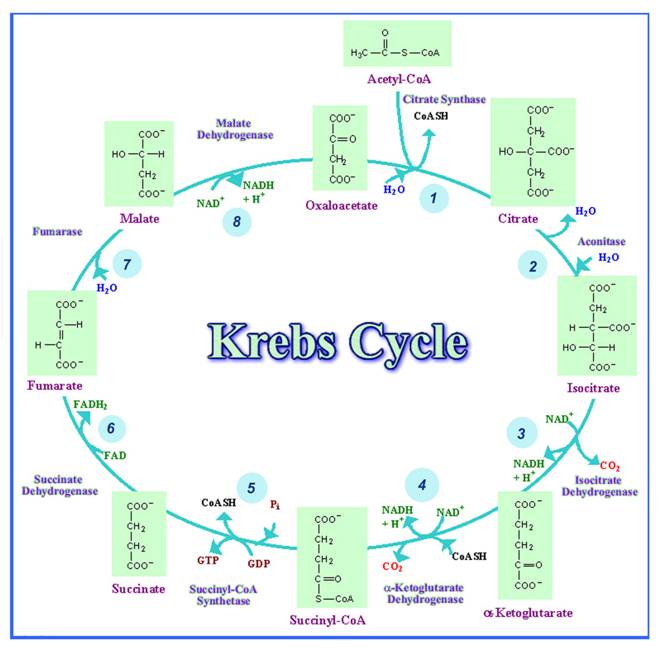
Step 1:
In the first step,
acetyl CoA is condensed with oxaloacetate to form citrate. The reaction is
catalyzed by the enzyme citrate synthase. This is reaction is aldol condensation
reaction and Citrate formation is highly exergonic because the standard free
energy change is equal to - 8 Kcal/mole.
Step 2:
In this step,
citrate reversibly converted to isocitrate by aconitase. During this
isomerization reaction, an intermediate called cis-aconitate is formed by
dehydration. The carbon-carbon double bond of cis-aconitate is then rehydrated
to form the isomer isocitrate. This reaction is carried out by aconitase
enzyme.
Step 3:
In this step,
oxidative decarboxylation of isocitrate, catalyzed by isocitrate dehydrogenase,
occurs in two steps. First, isocitrate is oxidized to form oxalosuccinate, a
transient intermediate. Immediate decarboxylation of oxalosuccinate results in
the formation of a- ketoglutarate.
There are two forms
of isocitrate dehydrogenase in mammals. The NAD+ requiring isozyme is found
only within mitochondria. The other isozyme, which requires NADP+, is found in
both the mitochondrial matrix and the cytoplasm. In some circumstances the
latter enzyme is used to within both compartments too generate NADPH, which is
required in biosynthetic processes.
Step 4:
In this step,
a- ketoglutarate is converted into
succinyl CoA by the enzyme a -
ketoglutarate dehydrogenase complex. This exergonic, oxidative decarboxylation
reaction is analogous to the pyruvate dehydrogenase reaction. In both
reactions, energy rich thioester molecules are products, that is, acetyl CoA and
Succinyl CoA. Other similarities between the two multienzyme complexes are that
the same cofactors (TPP, COA-SH, Lipoic acid, NAD+ and FAD) are required and the
same or similar allosteric effectors are
inhibitors. In the case of a- ketoglutarate dehydrogenase, inhibition
is produced by succinyl CoA, NADH, ATP and GTP. An important difference
between the two complexes is that the control mechanism of a-
ketoglutarate dehydrogenase does not involve covalent modification.
Step 5:
In this step,
Succinyl CoA is converted into Succinate with the formation of GTP through
substrate level phosphorylation process. This reaction is catalyzed by
succinate thiokinase. In mammals GDP is phosphorylated whereas in many other
organisms, ADP is phosphorylated instead.
Step 6:
In this step,
succinate dehydrogenase catalyzes the oxidation of succinate to form fumarate.
Unlike the other citric acid cycle enzymes, succinate dehydrogenase is not found
within the mitochondrial matrix. Instead, it is tightly bound to the inner
mitochondrial membrane. Succinate dehydrogenase is activated by high
concentrations of succinate, ATP, and Pi and inhibited by oxaloacetate. This
enzyme also inhibited by malonate which is an structural analog of succinate.
Step 7:
In this step,
fumarate is converted to L-malate in a reversible stereospecific hydration
reaction catalyzed by fumarase which is also otherwise known as fumarate
hydratase.
Step 8:
In the final step,
oxaloacetate is regenerated with the oxidation of L-malate by the enzyme malate
dehydrogenase. Malate dehydrogenase uses NAD+ as the oxidizing agent in a
highly endergonic reaction.
REGULATION OF TCA :
The cirtric acid
cycle enzymes citrate synthase, isocitrate dehydrogenase and
a- ketoglutarate dehydrogenase are
closely regulated because they catalyze reactions that represent important
metabolic branch points.
Citrate synthase:
This enzyme is
regulated by the concentration of substrate oxaloacetate and acetyl CoA.
Because of acetyl CoA and oxaloacetate are low in mitochondria in relation to
the amount of the enzyme, any increase in substrate availability stimulates
citrate synthesis. High concentrations of succinyl CoA and citrate inhibit
citrate synthase by acting as allosteric inhibitors. Other allosteric
regulators of this reaction are NADH and ATP, whose concentrations reflect the
cell’s current energy status. A resting cell has high NADH / NAD + and ATP /
ADP ratios. As a cell becomes metabolically active, NADH and ATP concentrations
decrease. Consequently, key enzymes such as citrate synthase become more
active.
Isocitrate Dehydrogenase:
Isocitrate
dehydrogenase catalyzes the second closely regulated reaction in the cycle. Its
activity is stimulated by relatively high concentrations of ADP and NAD + and
inhibited by ATP and NADH. This enzyme is closely regulated because of its
important role in citrate metabolism.
a
- ketoglutarate Dehydrogenase:
This enzyme is
strictly regulated because of the important role of
a - ketoglutarate in several metabolic
processes. When a cell’s energy stores are low, of
a - ketoglutarate dehydrogenase is
activated and of a - ketoglutarate is
retained within the cycle at the expense of biosynthetic processes. As the
cell’s supply of NADH rises, the enzyme is inhibited and of
a - ketoglutarate molecules become
available for biosynthetic reactions.
Fluroacetate can
inhibit aconitase and then inhibit TCA cycle.
TCA IS AMPHIOBOLIC:
The citric acid
cycle is obviously catabolic, since acetyl groups are oxidized to form CO2 and
energy is conserved in reduced coenzyme molecules. The citric acid cyle is also
anabolic, since several citric acid cycle intermediates are precursors in
biosynthetic pathway. For example, oxaloacetate is used in both gluconeogenesis
and aminoacid synthesis. a -
ketoglutarate also plays an important role in aminoacid synthesis. The
synthesis of porphyrins such as heme uses succinyl CoA. Finally, the synthesis
of fatty acids and cholesterol in the cytoplasm requires acetyl CoA. Because
acetyl CoA cannot penetrate in the inner mitochondrial membrane, it is converted
to citrate. After its transport into the cytoplasm, citrate is cleaved to form
acetyl CoA and oxaloacetate by citrate lyase.
Anaplerotic
reaction, a reaction that replenishes a substrate needed for a biochemical
pathway, available to replenish the intermediates of citric acid cycle. One of
the most important anaplerotic reactions is catalyzed by pyruvate carboxylase.
A high concentration of acetyl CoA, an indicator of an insufficient oxaloacetate
concentration, activates pyruvate carboxylase. As a result, oxaloacetate
concentration increases. Any excess oxaloacetate that is not used within the
citric acid cycle is used in gluconeogenesis. Other anaplerotic reactions
include the synthesis of succinyl CoA from certain fatty acids and certain amino
acids.
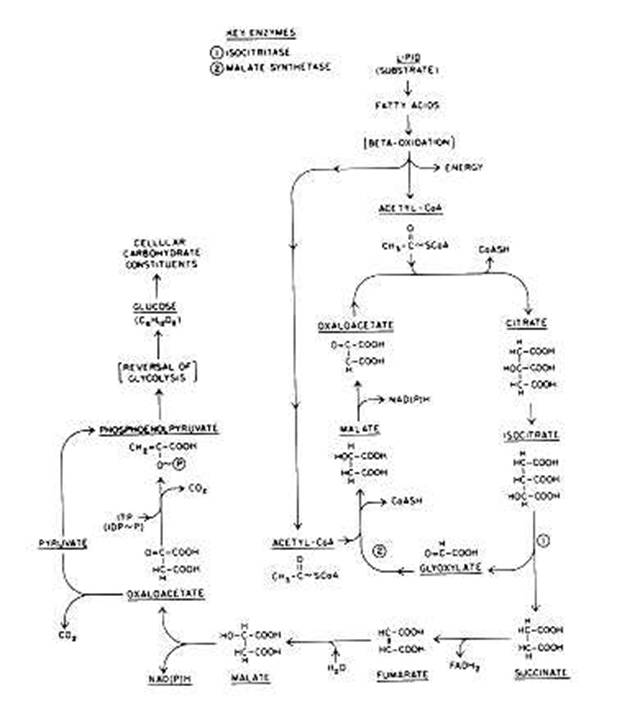
Glyoxylate Cycle :
In general, the Krebs cycle functions
similarly in bacteria and eukaryotic systems, but major differences are found
among bacteria. One difference is that in obligate aerobes, L-malate may be
oxidized directly by molecular 02 via an electron transport chain. In
other bacteria, only some Krebs cycle intermediate reactions occur because
a-ketoglutarate dehydrogenase is missing.
A modification of the Krebs cycle,
commonly called the glyoxylate cycle, or shunt , which exists in some bacteria.
This shunt functions similarly to the Krebs cycle but lacks many of the Krebs
cycle enzyme reactions. The glyoxylate cycle is primarily an oxidative pathway
in which acetyl~SCoA is generated from the oxidation, of acetate, which usually
is derived from the oxidation of fatty acids. The oxidation of fatty acids to
acetyl~SCoA is carried out by the b-oxidation pathway. Pyruvate oxidation is not
directly involved in the glyoxylate shunt, yet this shunt yields sufficient
succinate and malate, which are required for energy production . The glyoxylate
cycle also generates other precursor compounds needed for biosynthesis . The
glyoxylate cycle was discovered as an unusual metabolic pathway during an
attempt to learn how lipid (or acetate) oxidation in bacteria and plant seeds
could lead to the direct biosynthesis of carbohydrates. The glyoxylate cycle
converts oxaloacetate either to pyruvate and CO2 (catalyzed by
pyruvate carboxylase) or to phosphoenolpyruvate and CO2 (catalyzed by
the inosine triphosphate [ITP]-dependent phosphoenolpyruvate carboxylase
kinase). Either triose compound can then be converted to glucose by reversal of
the glycolytic pathway. The glyoxylate cycle is found in many bacteria,
including Azotobacter vinelandii and particularly in organisms that grow
well in media in which acetate and other Krebs cycle dicarboxylic acid
intermediates are the sole carbon growth source. One primary function of the
glyoxylate cycle is to replenish the tricarboxylic and dicarboxylic acid
intermediates that are normally provided by the Krebs cycle. A pathway whose
primary purpose is to replenish such intermediate compounds is called
anaplerotic.