RESPIRTORY SYSTEM
Organization of the Respiratory System
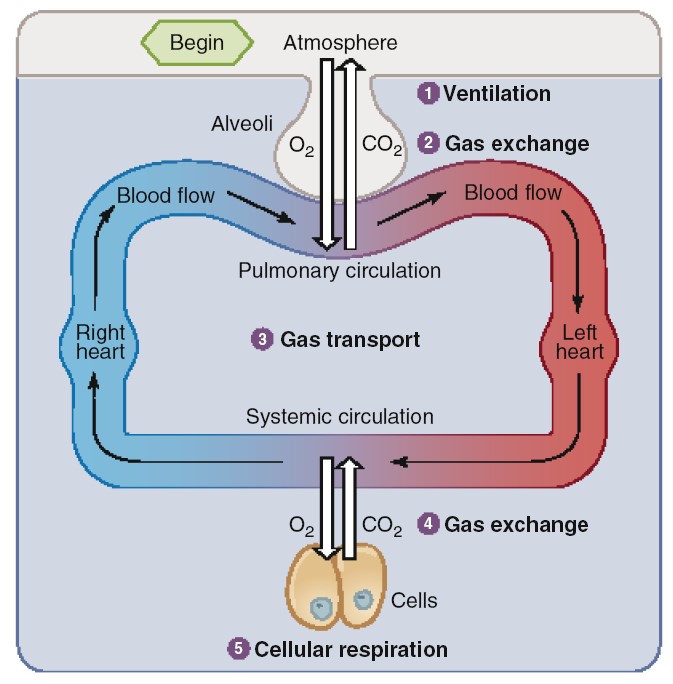
Respiration is defined as the movement of oxygen from the outside environment to
the cells within tissue and the transport of carbon dioxide in the opposite
direction. There are two lungs, the
right and left, each divided into lobes. The lungs consist mainly of tiny
air-containing sacs called alveoli (singular, alveolus), which
number approximately 300 million in an adult. The alveoli are the sites of gas
exchange with the blood. The airways are the tubes through which air
flows from the external environment to the alveoli and back. Inspiration
(inhalation) is the movement of air from the external environment through the
airways into the alveoli during breathing. Expiration (exhalation) is air
movement in the opposite direction. An inspiration and expiration constitute a
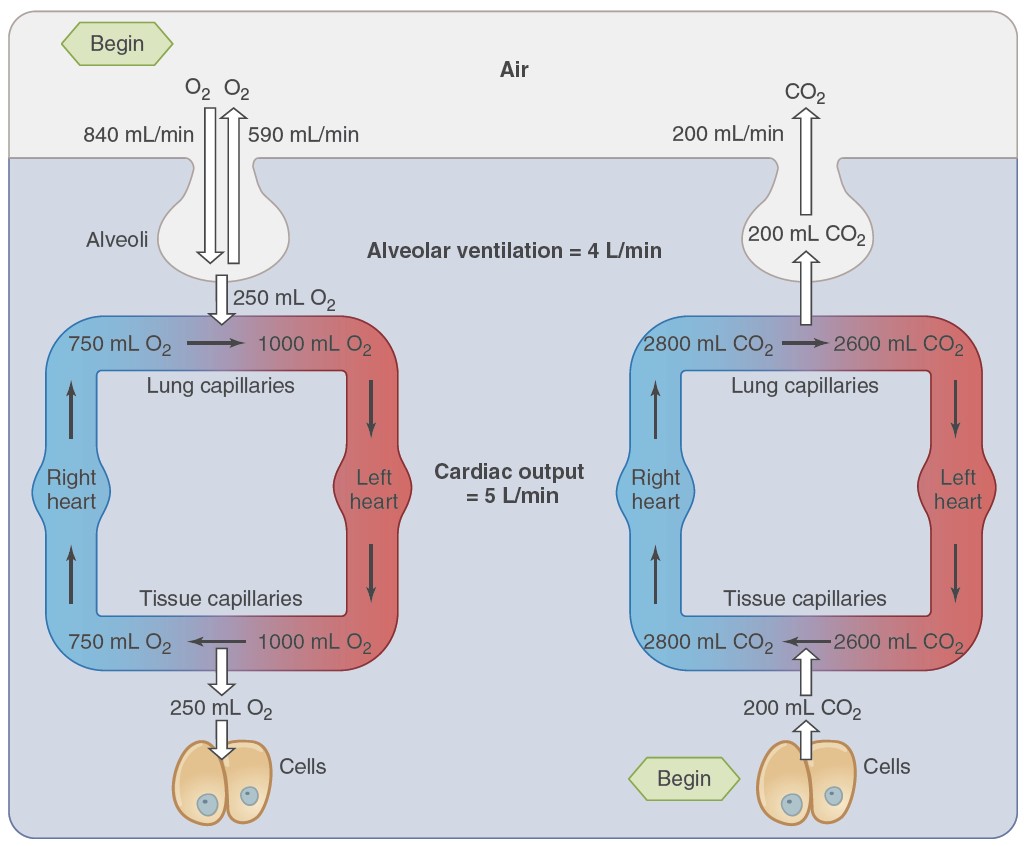
respiratory cycle. During the entire respiratory cycle, the right
ventricle of the heart pumps blood through the pulmonary arteries and arterioles
and into the capillaries surrounding each alveolus. In a healthy adult at rest,
approximately 4 L of fresh air enters and leaves the alveoli per minute, while
5L of blood, the cardiac output, flows through the pulmonary capillaries. During
heavy exercise, the airflow can increase 20-fold, and the blood flow five to six
fold.

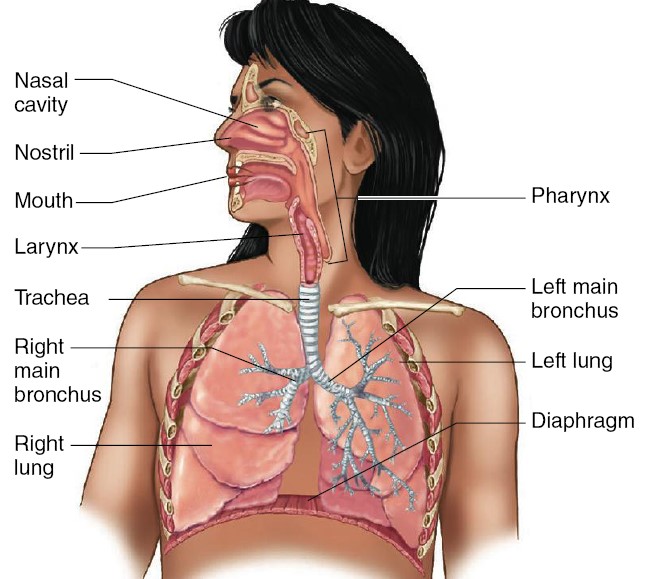
The Airways and Blood Vessels
During inspiration, air passes through the nose or the mouth (or both) into the
pharynx, a passage common to both air and food. The pharynx branches into
two tubes: the esophagus, through which food passes to the stomach, and the
larynx, which is part of the airways. The larynx houses the vocal cords,
two folds of elastic tissue stretched horizontally across its lumen. The
flow of air past the vocal cords causes them to vibrate, producing sounds. The
nose, mouth, pharynx, and larynx are collectively termed the upper airways.
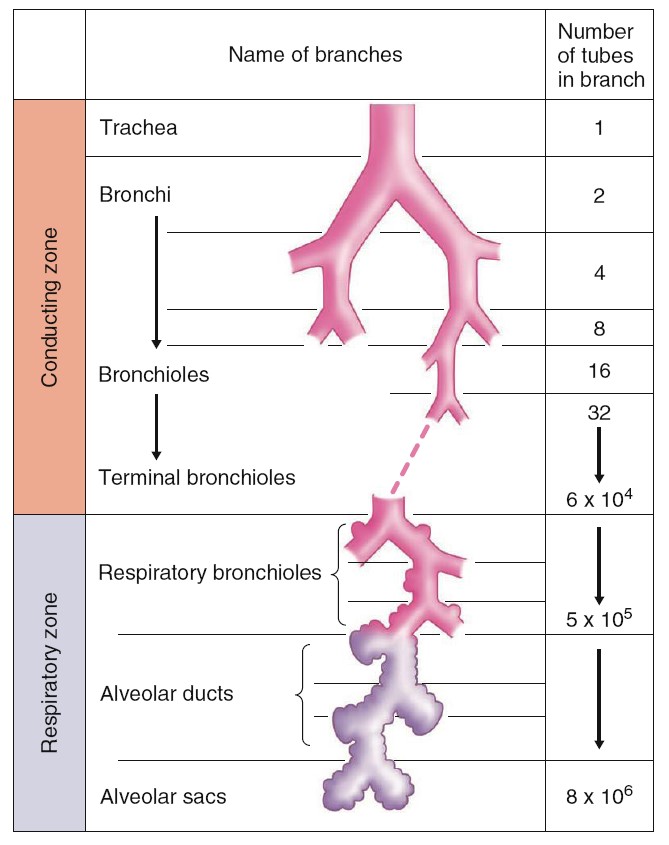
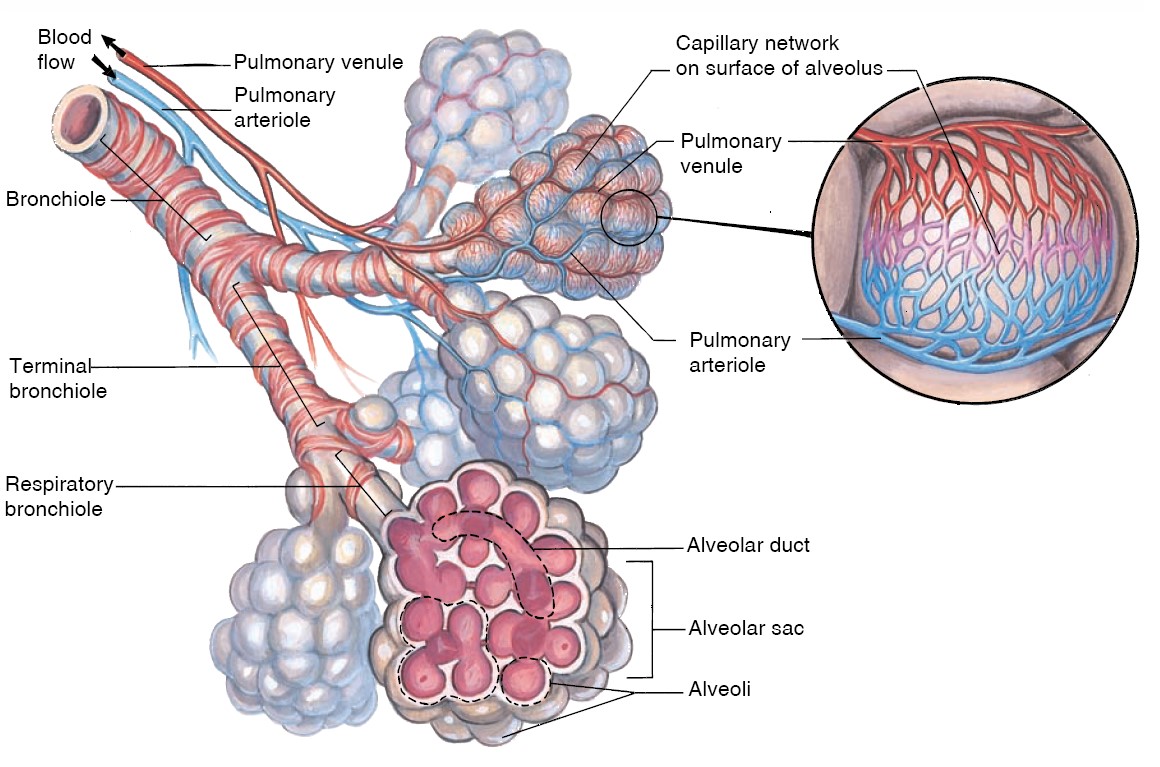
The larynx opens into a long tube, the trachea, which in turn
branches into two bronchi (singular, bronchus), one of which
enters each lung. Within the lungs, there are more than 20 generations of
branchings, each resulting in narrower, shorter, and more numerous tubes. The
walls of the trachea and bronchi contain rings of cartilage, which give them
their cylindrical shape and support them. The first airway branches that no
longer contain cartilage are termed bronchioles, which branch into the
smaller, terminal bronchioles. Alveoli first begin to appear attached to the
walls of the respiratory bronchioles. The number of alveoli increases
in the alveolar ducts, and the airways then end in grapelike clusters called
alveolar sacs that consist entirely of alveoli. The bronchioles are
surrounded by smooth muscle, which contracts or relaxes to alter bronchiolar
radius, in much the same way that the radius of small blood vessels (arterioles)
is controlled. The airways beyond the larynx can be divided into two zones. The
conducting zone extends from the top of the trachea to the end of the
terminal bronchioles. This zone contains no alveoli and does not exchange gases
with the blood. The respiratory zone extends from the respiratory
bronchioles down. This zone contains alveoli and is the region where gases
exchange with the blood.
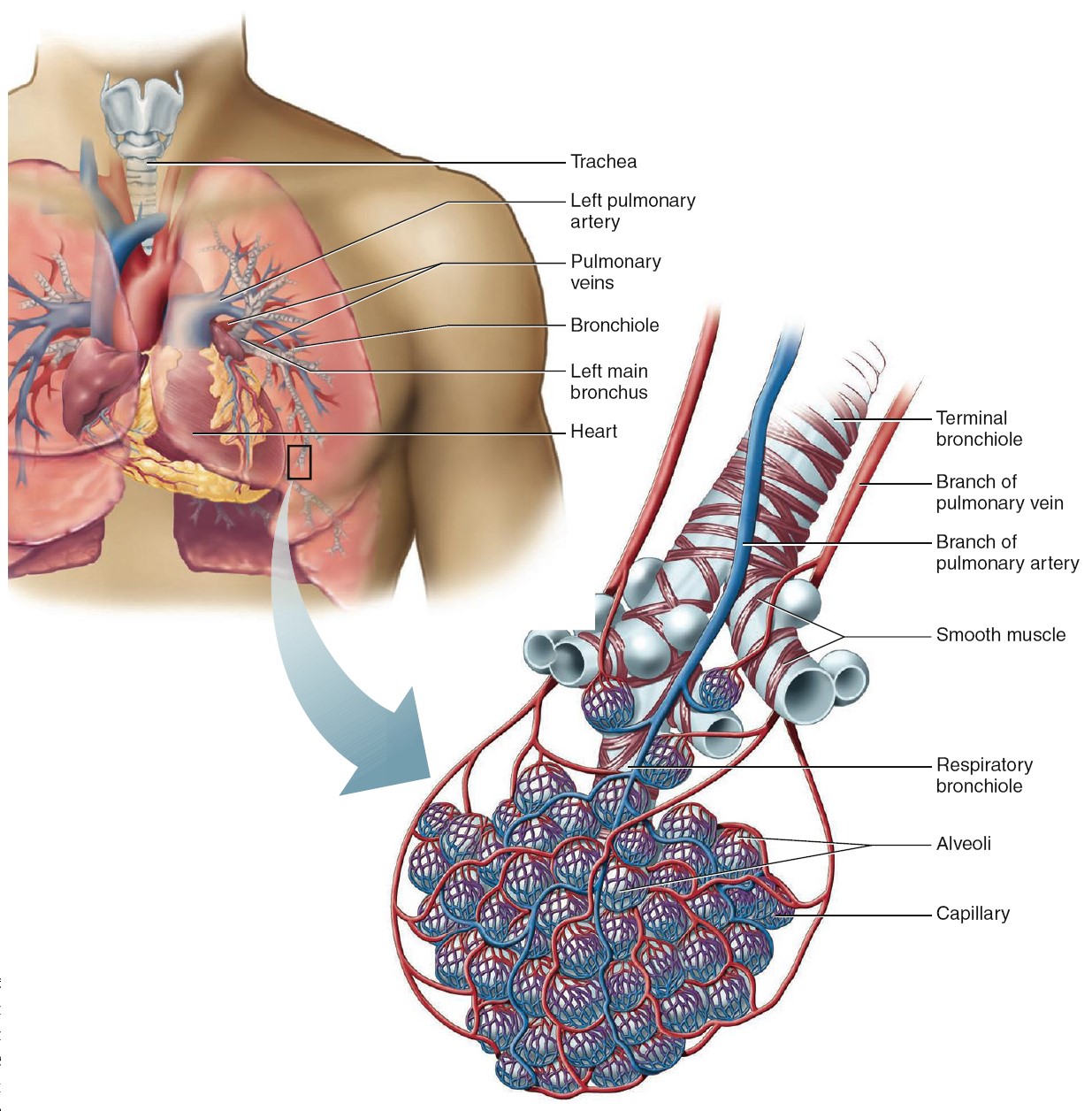
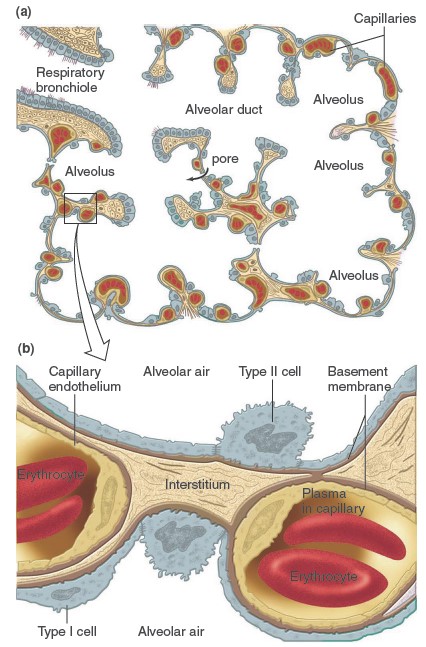
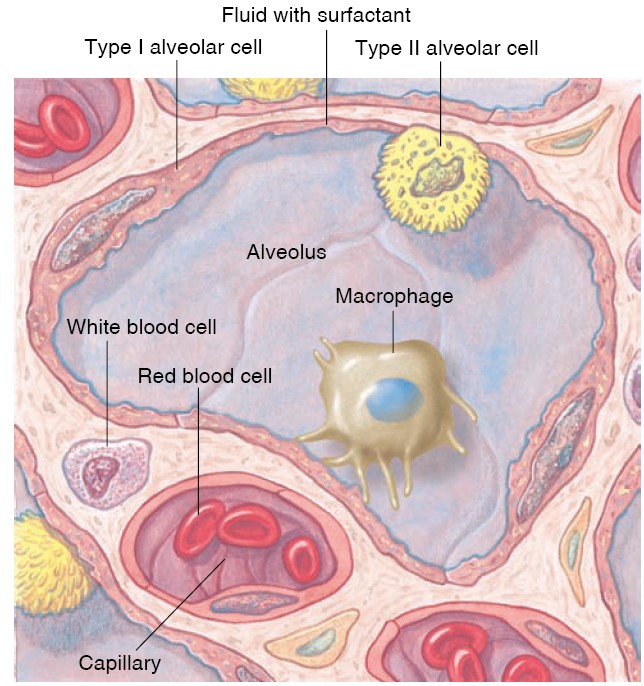
Site of Gas Exchange: The Alveoli
The alveoli are tiny, hollow sacs with open ends that are continuous with the
lumens of the airways. Typically, a single alveolar wall separates the air in
two adjacent alveoli. Most of the air-facing surfaces of the wall are lined by a
continuous
layer, one cell thick, of flat epithelial cells termed type I
alveolar cells. Interspersed between these cells are thicker, specialized
cells termed type II alveolar cells that
produce a detergent-like substance called surfactant that, is important for
preventing the collapse of the alveoli. The alveolar walls contain capillaries
and a very small interstitial space, which consists of interstitial fluid and a
loose meshwork of connective tissue. In many places, the interstitial space is
absent altogether, and the basement membranes of the alveolar-surface epithelium
and the capillary-wall endothelium fuse. Because of this unique anatomical
arrangement, the blood within an alveolar-wall capillary is separated from the
air within the alveolus by an extremely thin barrier (0.2 mm, compared with the
7mm diameter of an average red blood cell). The total surface area
of alveoli in contact with capillaries is roughly the size of a tennis court.
This extensive area and the thinness of the barrier permit the rapid exchange of
large quantities of oxygen and carbon dioxide by diffusion. These are excellent
examples of two of the general principles of physiology—that physiological
processes require the transfer and balance of matter (in this case, oxygen and
carbon dioxide) and energy between compartments; and that structure (in this
case, the thinness of the diffusion barrier a nd the enormous surface area for
gas exchange) is a determinant of—and has coevolved with—function (the transfer
of oxygen and carbon dioxide between the alveolar air and the blood in the
pulmonary capillaries). In some of the alveolar walls, pores permit the flow of
air between alveoli. This route can be very important when the airway leading to
an alveolus is occluded by disease, because some
air can still enter the alveolus by way of the pores between it and adjacent
alveoli.
COMPOSITIONS OF ALVEOLAR AIR AND
ATMOSPHERIC AIR ARE DIFFERENT
Alveolar air does not have the same concentrations of gases as atmospheric air.
There are several reasons for the differences. First, alveolar air is only
partially replaced by atmospheric air with each breath. Second, O2 is constantly
being absorbed into the pulmonary blood from the alveolar air. Third, CO2 is
constantly diffusing from the pulmonary blood into the alveoli. And fourth, dry
atmospheric air that enters the respiratory passages is humidified even before
it reaches the alveoli. By volume, dry air contains 78.09% nitrogen, 20.95%
oxygen, 0.93% argon, 0.03% carbon dioxide and small amounts of other gases.
Nitrogen is a naturally occurring element that is essential for growth and
reproduction of both animal and plants.
It is found in amino acids, nucleic acid and biologically active
molecules like alkaloids. Most of
the living organisms cannot utilize elemental nitrogen directly but they can
utilized nitrogen fixed compounds. Nitrogen fixation achieved through physical
activity of lightning or biological activity of microbes.
Animal metabolism of nitrogen in proteins excreted as urea whereas in nucleic
acid excreted as urea and uric acid.
The characteristic odor of animal flesh decay is caused by the creation of long
chain nitrogen containing amines such as putrescine and cadaverine which are
breakdown product of ornithine and lysine respectively.
Most decay eventually return nitrogen to the atmosphere.
Oxygen plays vital role in the breathing process and in the metabolism of the
living organisms. Most of the
organic compounds of living organisms contains oxygen in fixed format.
During breathing, oxygen enter into human and it is transported through
blood to tissue and then it reaches cells.
In cells, oxygen plays vital role in cell respiration process in which
oxygen converted into water.
HUMIDIFICATION OF THE AIR IN THE RESPIRATORY PASSAGES
Atmospheric air is composed almost entirely of nitrogen and O2; it normally
contains almost no CO2 and little water vapor. However, as soon as the
atmospheric air enters the respiratory passages, it is exposed to the fluids
that cover the respiratory surfaces. Even before the air enters the alveoli, it
becomes almost totally humidified. The partial pressure of water vapor at a
normal body temperature of 37°C is 47 mm Hg, which is therefore the partial
pressure of water vapor in the alveolar air. Because the total pressure in the
alveoli cannot rise to more than the atmospheric pressure (760 mm Hg at sea
level), this water vapor simply dilutes all the other gases in the
inspired air. Humidification of the air dilutes the oxygen partial pressure at
sea level from an average of 159 mm Hg in atmospheric air to 149 mm Hg in the
humidified air, and it dilutes the nitrogen partial pressure from 597 to 563 mm
Hg.
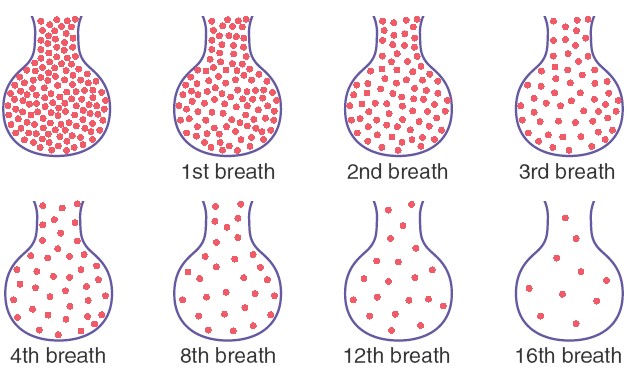
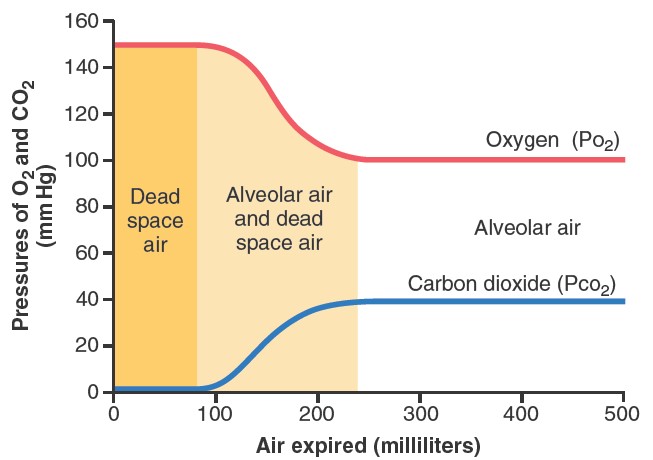
ALVEOLAR AIR IS SLOWLY RENEWED BY ATMOSPHERIC AIR
The average male functional residual capacity of the lungs (the volume of
air remaining in the lungs at the end of normal expiration) measures about 2300
milliliters. Yet only 350 milliliters of new air is brought into the alveoli
with each normal inspiration, and this same amount of old alveolar air is
expired. Therefore, the volume of alveolar air replaced by new atmospheric air
with each breath is only one seventh of the total, so multiple breaths are
required to exchange most of the alveolar air. This slows rate of renewal of the
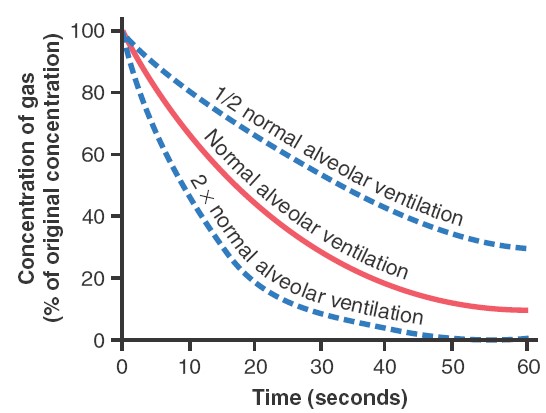
alveolar air. In the first alveolus of the figure, excess gas is present in the
alveoli but note that even at the end of 16 breaths the excess gas still has not
been completely removed from the alveoli. The graph showed that the rate at
which excess gas in the alveoli is normally removed, showing that with normal
alveolar ventilation, about one half the gas is removed in 17 seconds. When a
person’s rate of alveolar ventilation is only one-half normal, one half the gas
is removed in 34 seconds, and when the rate of ventilation is twice normal, one
half is removed in about 8 seconds.
Importance of the Slow Replacement of Alveolar Air.
The slow replacement of alveolar air is of particular importance in preventing
sudden changes in gas concentrations in the blood. This makes the respiratory
control mechanism much more stable than it would be otherwise, and it helps
prevent excessive increases and decreases in tissue oxygenation, tissue CO2
concentration,
and tissue pH when respiration is temporarily interrupted.
Boyle’s Law
Changes in intrapulmonary pressure occur as a result of changes in lung volume.
This follows from Boyle’s law, which states that the pressure of a given
quantity of gas is inversely proportional to its volume. An increase in lung
volume during inspiration decreases intrapulmonary pressure to subatmospheric
levels; air therefore goes in. A decrease in lung volume, conversely, raises the
intrapulmonary pressure above that of the atmosphere, expelling air from the
lungs. These changes in lung volume occur as a consequence of changes in
thoracic volume.
According to Dalton’s law, the total pressure of a gas mixture (such as
air) is equal to the sum of the pressures that each gas in the mixture would
exert independently. The pressure that a particular gas in a mixture exerts
independently is the partial pressure of that gas, which is equal to the
product of the total pressure and the fraction of that gas in the mixture.
Dalton’s law can thus be restated as follows: The total pressure of the gas
mixture is equal to the sum of the partial pressures of the constituent gases.
Because oxygen constitutes about 21% of the atmosphere, for example, its partial
pressure (abbreviated PO2) is 21% of 760, or about 159 mmHg. Nitrogen
constitutes about 78% of the atmosphere, so its partial pressure is equal to
0.78. 760 = 593 mmHg. These two gases thus contribute about 99% of the total
pressure of 760 mmHg:
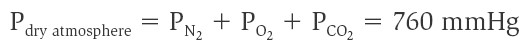
Partial Pressures of Gases in Blood
The enormous surface area of alveoli and the short diffusion distance between
alveolar air and the capillary blood quickly help to bring oxygen and carbon
dioxide in the blood and air into equilibrium. This function is further aided by
the tremendous number of capillaries that surround each alveolus, forming an
almost continuous sheet of blood around the alveoli. When a liquid and a gas,
such as blood and alveolar air, are at equilibrium, the amount of gas dissolved
in the fluid reaches a maximum value. According to Henry’s law, this
value depends on (1) the solubility of the gas in the fluid, which is a physical
constant; (2) the temperature of the fluid—more gas can be dissolved in cold
water than warm water; and (3) the partial pressure of the gas. Because
solubility is a constant and the temperature of the blood does not vary
significantly, the concentration of a gas dissolved in a fluid (such as
plasma) depends directly on its partial pressure in the gas mixture. When
water—or plasma—is brought into equilibrium with air at a PO2
of 100 mmHg, for example, the fluid will contain 0.3 ml of O2
per 100 ml fluid at 37 ° C. If the PO2
of the gas were reduced by half, the amount of dissolved oxygen would also be
reduced by half.
OXYGEN CONCENTRATION AND PARTIAL PRESSURE IN THE ALVEOLI
Oxygen is continually being absorbed from the alveoli into the blood of the
lungs, and new O2 is continually being breathed into the alveoli from the
atmosphere. The more rapidly O2 is absorbed, the lower its concentration in the
alveoli becomes; conversely, the more rapidly new O2 is breathed into the
alveoli from the atmosphere, the higher its concentration becomes. Therefore, O2
concentration in the alveoli, as well as its partial pressure, is controlled by
(1) the rate of absorption of O2 into the blood and (2) the rate of entry of new
O2 into the lungs by the ventilatory process.
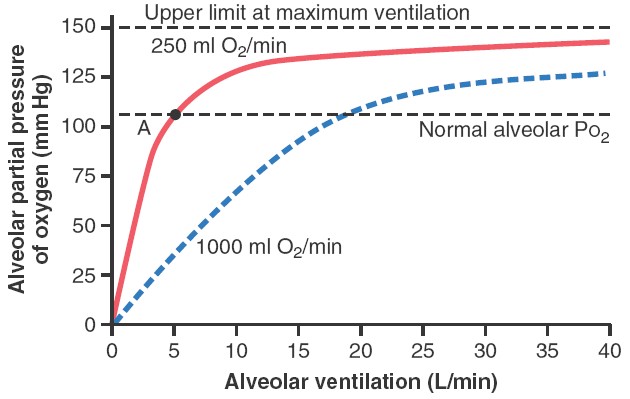
The figure
shows the effect of alveolar ventilation and rate of O2 absorption into the
blood on the alveolar partial pressure of O2 (PO2). One curve represents O2
absorption at a rate of 250 ml/min, and the other curve represents a rate of
1000 ml/min. At a normal ventilatory rate of 4.2 L/min and an O2 consumption of
250 ml/min, the normal operating point in figure is point A. The figure also
shows that when 1000 milliliters of O2 are being absorbed each minute, as occurs
during moderate exercise, the rate of alveolar ventilation must increase
fourfold to maintain the alveolar Po2 at the normal value of 104 mm Hg. Another
effect shown in figure is that even an extreme increase in alveolar ventilation
can never increase the alveolar PO2 above 149 mm Hg as long as the person is
breathing normal atmospheric air at sea level pressure, because 149 mm Hg is the
maximum PO2 in humidified air at this pressure. If the person breathes gases
that contain partial pressures of O2 higher than 149 mm Hg, the alveolar PO2 can
approach these higher pressures at high rates of ventilation.
CO2 CONCENTRATION AND PARTIAL PRESSURE IN THE ALVEOLI
Carbon dioxide is continually formed in the body and then carried in the blood
to the alveoli, and it is continually removed from the alveoli by ventilation.
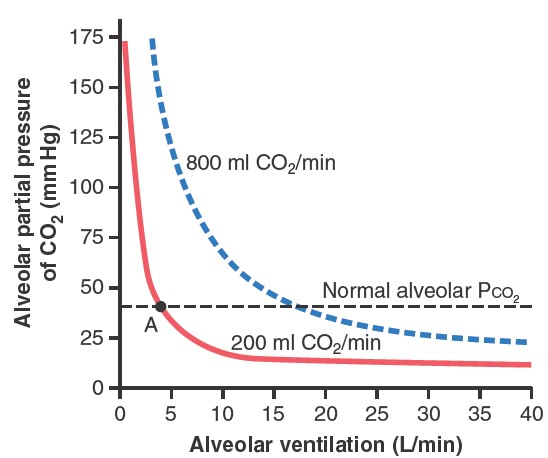
The figure
shows the effects on the alveolar partial pressure of CO2 (PCO2) of
both alveolar ventilation and two rates of CO2
excretion, 200 and 800 ml/min. One curve represents a normal rate of CO2
excretion of 200 ml/min. At the normal rate of alveolar ventilation of 4.2
L/min, the operating point for alveolar PCO2 is at point A in figure (i.e., 40
mm Hg). Two other facts are also evident from figure:
First, the alveolar PCO2 increases directly in proportion to the rate of CO2
excretion, as represented by the fourfold elevation of the curve (when 800
milliliters of CO2 are excreted per minute). Second, the alveolar PCO2
decreases
in inverse proportion to alveolar ventilation. Therefore,
the concentrations and partial pressures of both O2 and CO2 in the alveoli are
determined by the rates of absorption or excretion of the two gases and by the
amount of alveolar ventilation.
Nitrogen Narcosis
Although at sea level nitrogen is physiologically inert, larger amounts of
dissolved nitrogen under hyperbaric (highpressure) conditions have deleterious
effects, possibly caused by the increased amounts of nitrogen dissolved in
plasma membranes at the high partial pressures. Nitrogen narcosis
resembles alcohol intoxication; depending on the depth of the dive, the diver
may experience what Jacques Cousteau termed “rapture of the deep.” Dizziness and
extreme drowsiness are other narcotizing effects.
Gas Exchange Between Alveoli and Blood
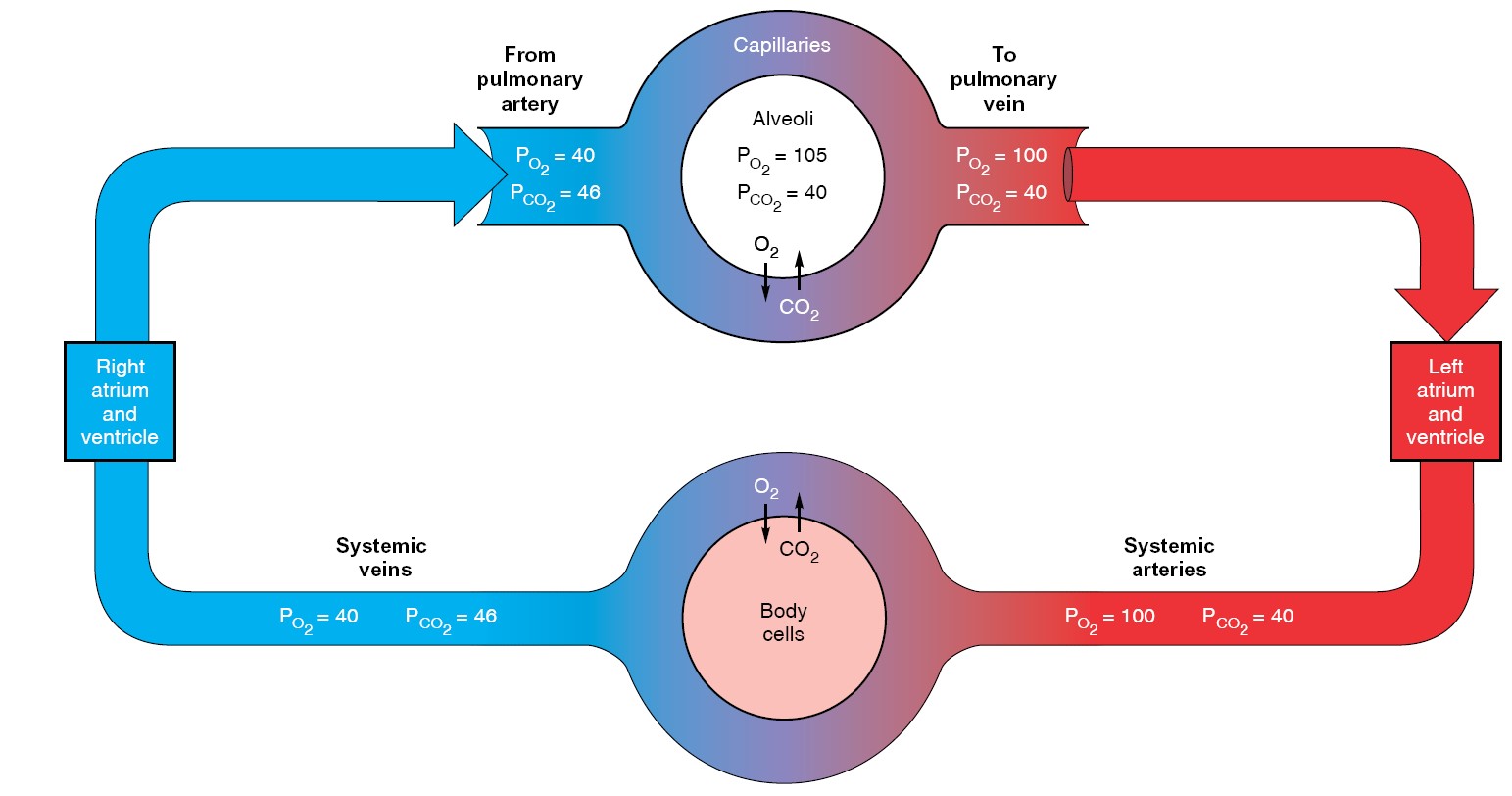
The blood that enters the pulmonary capillaries is systemic venous blood pumped
by the right ventricle to the lungs through the pulmonary arteries. Having come
from the tissues, it has a relatively high PCO2 (46 mmHg in a healthy
person at rest) and a relatively
low PO2
(40 mmHg).
The differences in the partial pressures of oxygen and carbon dioxide on the two
sides of the alveolar-capillary membrane result in the net diffusion of oxygen
from alveoli to blood and of carbon dioxide from blood to alveoli. (For
simplicity, we are ignoring the small diffusion barrier provided by the
interstitial space.) As this diffusion occurs, the PO2 in the pulmonary
capillary blood increases and the PCO2 decreases. The net diffusion of
these gases ceases when the capillary partial pressures become equal to those in
the alveoli. In a healthy person, the rates at which oxygen and carbon dioxide
diffuse are high enough and the blood flow through the capillaries slow enough
that complete equilibrium is reached well before the blood reaches the end of
the capillaries.
Thus, the blood that leaves the pulmonary capillaries to return to the heart and
be pumped into the systemic arteries has essentially the same PO2 and
PCO2 as alveolar air. (They are not exactly the same, for reasons given
later.) Accordingly, the factors described in the previous section—atmospheric
PO2, cellular oxygen consumption and carbon dioxide production, and
alveolar ventilation—determine the alveolar gas pressures, which then determine
the systemic arterial gas pressures.
The diffusion of gases between alveoli and capillaries may be impaired in a
number of ways, resulting in inadequate oxygen diffusion into the blood. For one
thing, the total surface area of all of the alveoli in contact with pulmonary
capillaries may be decreased. In pulmonary edema, some of the
alveoli may become filled with fluid. Diffusion may also be impaired if the
alveolar walls become severely thickened with connective tissue (fibrotic), as,
for example, in the disease called diffuse interstitial fibrosis.
In this disease, fibrosis may arise from infection, autoimmune disease,
hypersensitivity to inspired substances, exposure to toxic airborne chemicals,
and many other causes. Typical symptoms of these types of diffusion diseases are
shortness of breath and poor oxygenation of blood. Pure diffusion problems of
these types are restricted to oxygen and usually do not affect the elimination
of carbon dioxide, which diffuses more rapidly than oxygen.
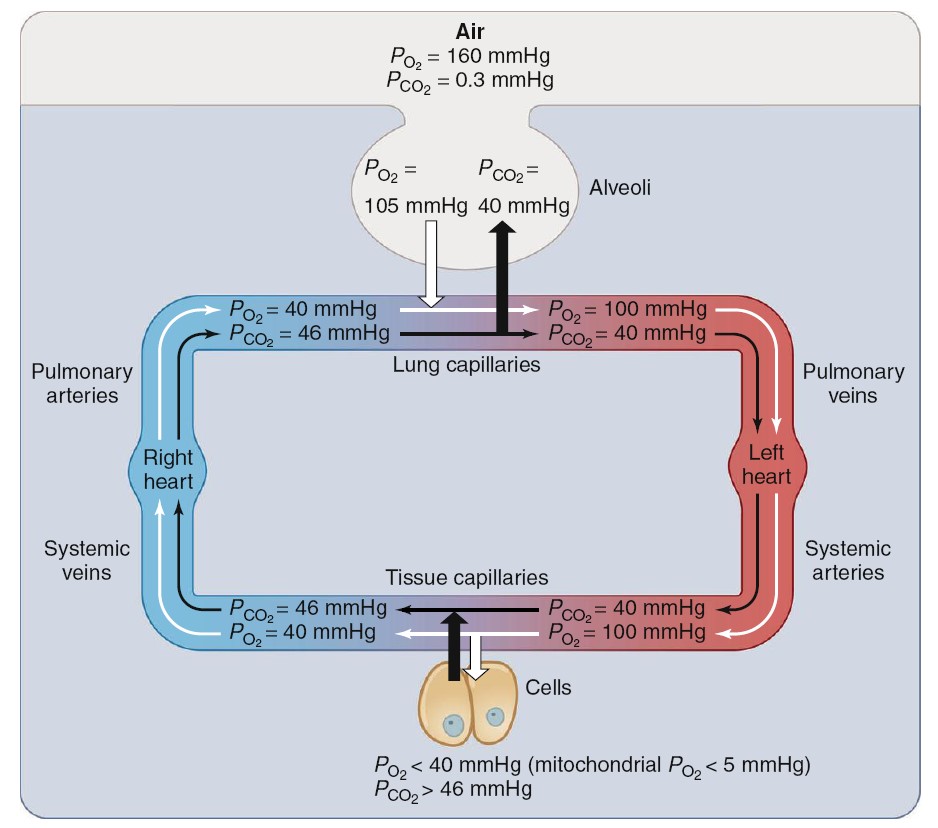
Gas Exchange between Tissues and Blood
As the systemic arterial blood enters capillaries throughout the body, it is
separated from the interstitial fluid by only the thin capillary wall, which is
highly permeable to both oxygen and carbon dioxide. The interstitial fluid, in
turn, is separated from the intracellular fluid by the plasma membranes of the
cells, which are also quite permeable to oxygen and carbon dioxide. Metabolic
reactions occurring within cells are constantly consuming oxygen and producing
carbon dioxide. Therefore, intracellular PO2 is lower and PCO2
higher than in arterial blood. The lowest PO2 of all—less than 5 mmHg—is
in the mitochondria, the site of oxygen utilization. As a result, a net
diffusion of oxygen occurs from blood into cells and, within the cells, into the
mitochondria, and a net diffusion of carbon dioxide occurs from cells into
blood. In this manner, as blood flows through systemic capillaries, its PO2
decreases and its PCO2 increases. In summary, the supply of new oxygen to
the alveoli and the consumption of oxygen in the cells create PO2
gradients that produce net diffusion of oxygen from alveoli to blood in the
lungs and from blood to cells in the rest of the body. Conversely, the
production of carbon dioxide by cells and its elimination from the alveoli via
expiration create PCO2 gradients that produce net diffusion of carbon
dioxide from cells to blood in the rest of the body and from blood to alveoli in
the lungs.
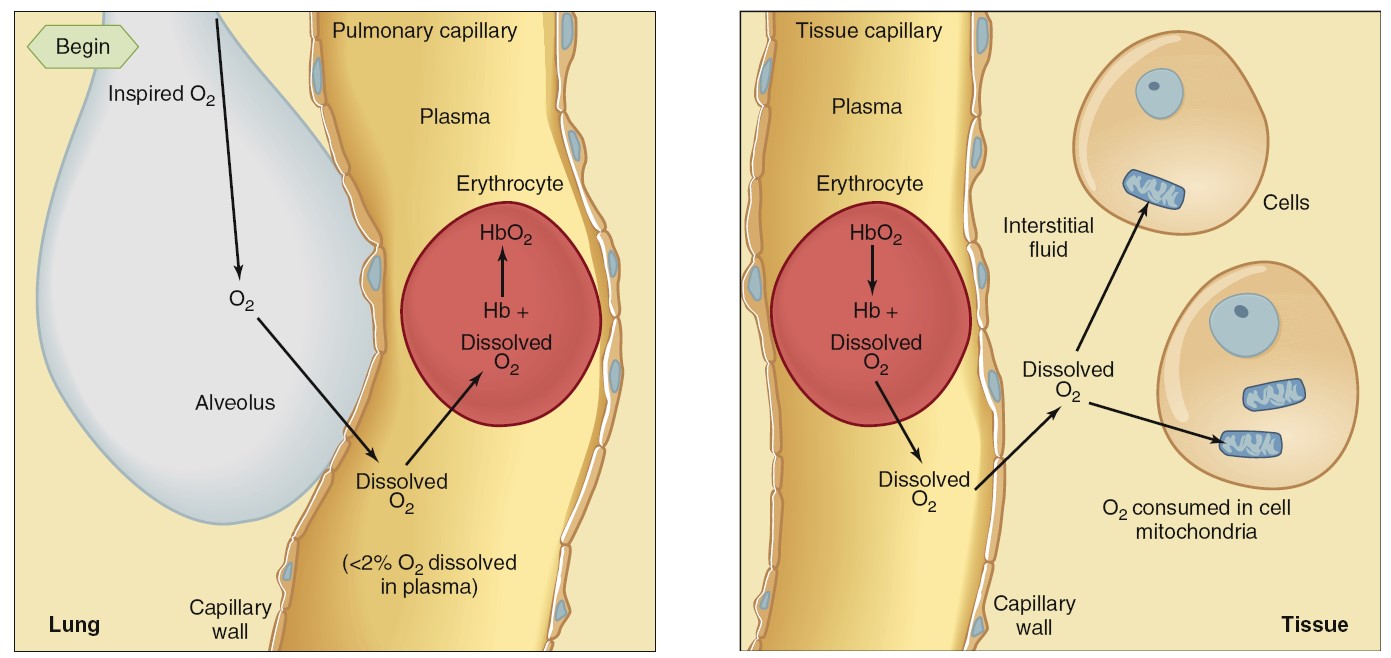
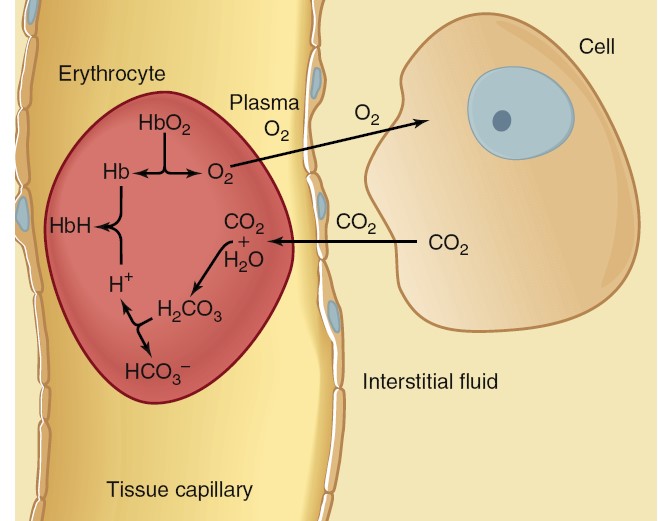
Transport of Oxygen in Blood
The oxygen is present in two forms: (1) dissolved in the plasma and erythrocyte
cytosol and (2) reversibly combined with hemoglobin molecules in the
erythrocytes. As predicted by Henry’s law, the amount of oxygen dissolved in
blood is directly proportional to the PO2 of the blood. Because the
solubility of oxygen in water is relatively low, only 3 mL can be dissolved in 1
L of blood at the normal arterial PO2 of 100 mmHg. The other 197 mL of
oxygen in a liter of arterial
blood—more than 98% of the oxygen content in the liter—is
transported in the erythrocytes, reversibly combined with hemoglobin. Each
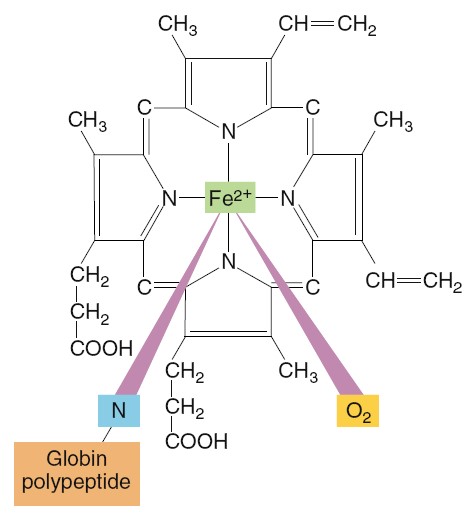
hemoglobin molecule is a protein made up of four subunits bound together.
Each subunit consists of a molecular group known as heme and a
polypeptide attached to the heme. The four polypeptides of a hemoglobin molecule
are collectively called globin. Each of the four heme groups in a
hemoglobin molecule
contains one atom of iron (Fe2+), to which molecular oxygen binds.
Because each iron atom can bind one molecule of oxygen, a single hemoglobin
molecule can bind four oxygen molecules. However, for simplicity, the equation
for the reaction between oxygen and hemoglobin is usually written in terms of a
single polypeptide–heme subunit of a hemoglobin molecule:

Therefore, hemoglobin can exist in one of two forms—deoxyhemoglobin (Hb)
and oxyhemoglobin (HbO2). In a blood sample containing many
hemoglobin molecules, the fraction of all the hemoglobin in the form of
oxyhemoglobin is expressed as the percent hemoglobin saturation:
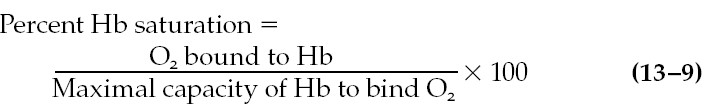
For example, if the amount of oxygen bound to hemoglobin is 40% of the maximal
capacity, the sample is said to be 40% saturated. The denominator in this
equation is also termed the oxygen-carrying capacity of the blood.
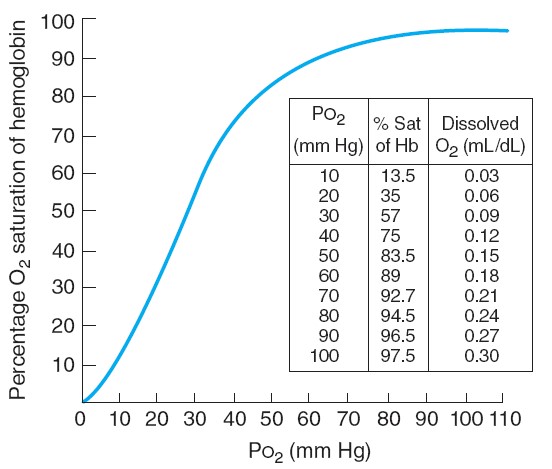
Let us now apply this analysis to capillaries of the lungs and tissues. The
plasma and erythrocytes entering the lungs have a PO2 of 40 mmHg. As we
can see from Figure 13.26, hemoglobin saturation at this PO2 is 75%. The
alveolar PO2-105 mmHg-is higher than the blood PO2 and so oxygen
diffuses from the alveoli into the plasma. This increases plasma
PO2 and induces diffusion of
oxygen into the erythrocytes, increasing erythrocyte PO2 and causing
increased combination of oxygen and hemoglobin. Most of the oxygen diffusing
into the blood from the alveoli does not remain dissolved but combines with
hemoglobin. Therefore, the blood PO2 normally remains less than the
alveolar PO2 until hemoglobin is virtually 100% saturated. This maintains
the diffusion gradient of oxygen movement into the blood during the very large
transfer of oxygen.
In the tissue capillaries, the process is reversed. Because the mitochondria of
all cells are utilizing oxygen, the cellular PO2 is less than the PO2
of the surrounding interstitial fluid. Therefore, oxygen is continuously
diffusing into the cells. This causes the interstitial fluid PO2 to
always be less than the PO2 of the blood flowing through the tissue
capillaries, so net diffusion of oxygen occurs from the plasma within the
capillary into the interstitial fluid. As a result, plasma PO2 becomes
lower than erythrocyte PO2, and oxygen diffuses out of the erythrocyte
into the plasma. The decrease in erythrocyte PO2 causes the dissociation
of oxygen from hemoglobin, thereby liberating oxygen, which then diffuses out of
the erythrocyte. The net result is a transfer, purely by diffusion, of large
quantities of oxygen from hemoglobin to plasma to interstitial fluid to the
mitochondria of tissue cells. In most tissues under resting conditions,
hemoglobin is still 75% saturated as the blood leaves the tissue capillaries.
This fact underlies an important local mechanism by which cells can obtain more
oxygen whenever they increase their activity. For example, an exercising muscle
consumes more oxygen, thereby lowering its intracellular and interstitial PO2.
This increases the blood-to-cell PO2 gradient. As a result, the rate of
oxygen diffusion from blood to cells increases. In turn, the resulting decrease
in erythrocyte PO2 causes additional dissociation of hemoglobin and
oxygen. In this manner, the extraction of oxygen from blood in an exercising
muscle is much greater than the usual 25%. In addition, an increased blood flow
to the muscles, called active hyperemia, also contributes greatly to the
increased oxygen supply.
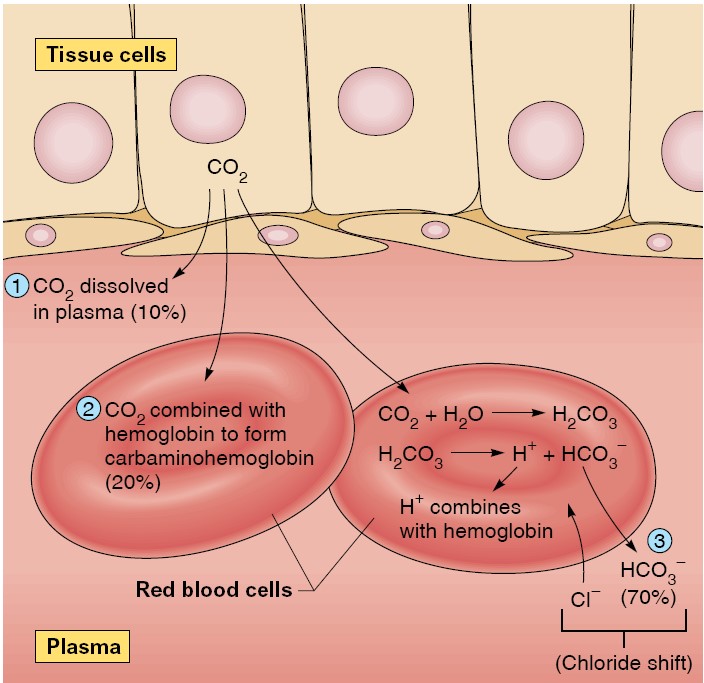
Transport of Carbon Dioxide in Blood
Carbon dioxide is a waste product that has toxicity in part because it generates
H+. Large changes in H+ concentration, if not buffered,
would lead to significant changes in pH, thus
altering
the tertiary structure of proteins, including enzymes. In a resting person,
metabolism generates about 200 mL of carbon dioxide per minute. When arterial
blood flows through tissue capillaries, this volume of carbon dioxide diffuses
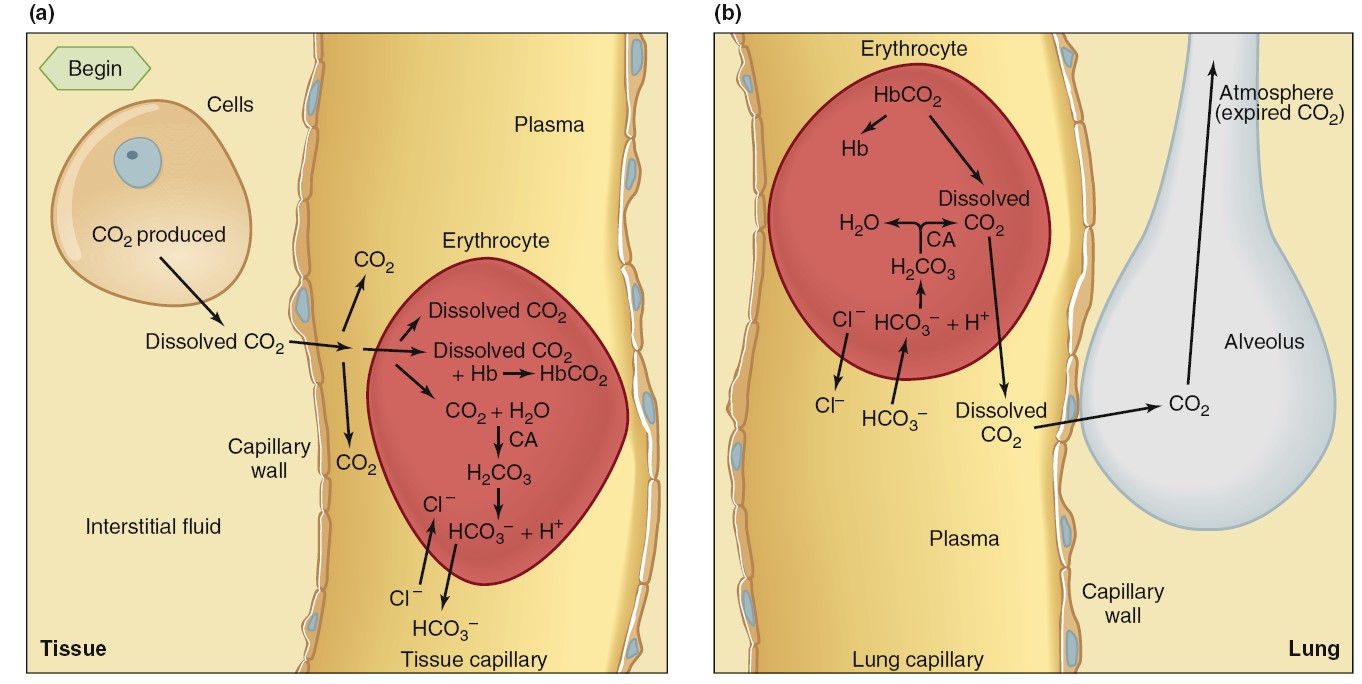
from the tissues into the blood. Carbon dioxide is much more soluble in water than is oxygen, so
blood carries more dissolved carbon dioxide than dissolved oxygen. Even so, only
about 10% of the carbon dioxide entering the blood dissolves in the plasma and
the cytosol of the erythrocytes. In order to transport all of the CO2 produced
in the tissues to the lung, much of the CO2 in the blood must be carried in
other forms. Another 25% to 30% of the carbon dioxide molecules entering the
blood react reversibly with the amino groups of hemoglobin to form
carbaminohemoglobin. For simplicity, this reaction with hemoglobin is
written as
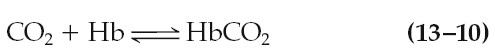
This reaction is aided by the fact that deoxyhemoglobin, formed as blood flows
through the tissue capillaries, has a greater affinity for carbon dioxide than
does oxyhemoglobin. The remaining 60% to 65% of the carbon dioxide molecules
entering the blood in the tissues is converted to HCO3-:
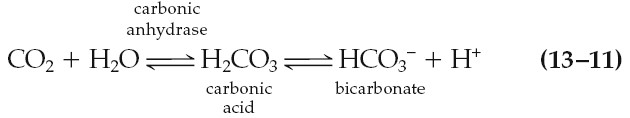
The first reaction in equation 13–11 is rate limiting and is very slow unless
catalyzed in both directions by the enzyme carbonic anhydrase. This
enzyme is present in the erythrocytes but not in the plasma, so this reaction
occurs mainly in the erythrocytes. In contrast, carbonic acid dissociates very
rapidly into HCO3- and H+ without any enzyme assistance.
Once formed, most of the HCO3- moves out of the erythrocytes into the
plasma via a transporter that exchanges one HCO3- for one chloride
ion (this is called the “chloride shift,” which maintains electroneutrality).
HCO3- leaving the erythrocyte favors the balance of the reaction
to the right.
The reactions also explain why, as mentioned earlier, the H+
concentration in tissue capillary blood and systemic venous blood is higher than
that in arterial blood and increases as metabolic activity increases. The fate
of this H+ will be discussed in the next section. Because carbon
dioxide undergoes these various fates in blood, it is customary to add up the
amounts of dissolved carbon dioxide, HCO3-, and carbon dioxide in
carbaminohemoglobin to arrive at the total-blood carbon dioxide, which is
measured as a component of routine blood chemistry testing. The opposite events
occur as systemic venous blood flows through the lung capillarie. Because the
blood PCO2 is higher than alveolar PCO2, a net diffusion of CO2
from blood into alveoli occurs. This loss of CO2 from the blood decreases the
blood PCO2 and drives the reactions to the left. HCO3- and H+
combine to produce H2CO3, which then dissociates to CO2 and H2O. Similarly,
HbCO2 generates Hb and free CO2. Normally, as fast as CO2 is generated from HCO3-
and H+ and from HbCO2, it diffuses into the alveoli. In this manner,
the CO2 that was delivered into the blood in the tissues is now delivered into
the alveoli, from where it is eliminated during expiration.
Transport of Hydrogen Ion Between Tissues and Lungs
As blood flows through the tissues, a fraction of oxyhemoglobin loses its oxygen
to become deoxyhemoglobin, while simultaneously a large quantity of carbon
dioxide enters the blood and undergoes the reactions that generate HCO3-
and H+. Deoxyhemoglobin
has a much greater affinity for H+ than does oxyhemoglobin, so it
binds (buffers) most of the H+. When deoxyhemoglobin binds H+,
it is abbreviated HbH.
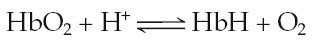
In this manner, only a small amount of the H+ generated in the blood
remains free. This explains why venous blood (pH 5 7.36) is only slightly more
acidic than arterial blood (pH 5 7.40). As the venous blood passes through the
lungs, this reaction is reversed. Deoxyhemoglobin becomes converted to
oxyhemoglobin and, in the process, releases the H+ it picked up in
the tissues. The H+ reacts with HCO3- to produce carbonic
acid, which, under the influence of carbonic anhydrase, dissociates to form
carbon dioxide and water. The carbon dioxide diffuses into the alveoli to be
expired. Normally, all the H+ that is generated in the tissue
capillaries from the reaction of carbon dioxide and water recombines with HCO3-
to form carbon dioxide and water in the pulmonary capillaries. Therefore, none
of this H+ appears in the arterial blood.
Not only would arterial PCO2 increase as a result, but so would arterial
H+ concentration. Increased
arterial H+ concentration due to
carbon dioxide retention is termed respiratory acidosis.
Conversely, hyperventilation would decrease arterial PCO2 and H+concentration,
producing respiratory alkalosis.
The Chloride Shift
As a result of catalysis by carbonic anhydrase within the red blood cells, large
amounts of carbonic acid are produced as blood passes through the systemic
capillaries. The buildup of carbonic acid concentrations within the red blood
cells favors the dissociation of these molecules into hydrogen ions (protons,
which contribute to the acidity of a solution) and HCO3−
(bicarbonate), as shown by this equation: H2 CO3 → H+ + HCO3−
The hydrogen ions (H+) released by the dissociation of carbonic acid are largely
buffered by their combination with deoxyhemoglobin within the red blood cells.
Although the unbuffered hydrogen ions are free to diffuse out of the red blood
cells, more bicarbonate diffuses outward into the plasma than does H+. As a
result of the “trapping” of hydrogen ions within the red blood cells by their
attachment to hemoglobin and the outward diffusion of bicarbonate, the inside of
the red blood cell gains a net positive charge. This attracts chloride ions
(Cl−), which move into the red blood cells as HCO3− moves out. This exchange of
anions as blood travels through the tissue capillaries is called the chloride
shift.
The unloading of oxygen is increased by the bonding of H+ (released from
carbonic acid) to oxyhemoglobin. This is the Bohr effect, and results in
increased conversion of oxyhemoglobin to deoxyhemoglobin. Now, deoxyhemoglobin
bonds H+ more strongly than does oxyhemoglobin, so the act of unloading its
oxygen improves the ability of hemoglobin to buffer the H+ released by carbonic
acid. Removal of H + from solution by combining with hemoglobin (through the law
of mass action), in turn, favors the continued production of carbonic acid and
thereby improves the ability of the blood to transport carbon dioxide. Thus,
carbon dioxide increases oxygen unloading, and oxygen unloading improves carbon
dioxide transport.
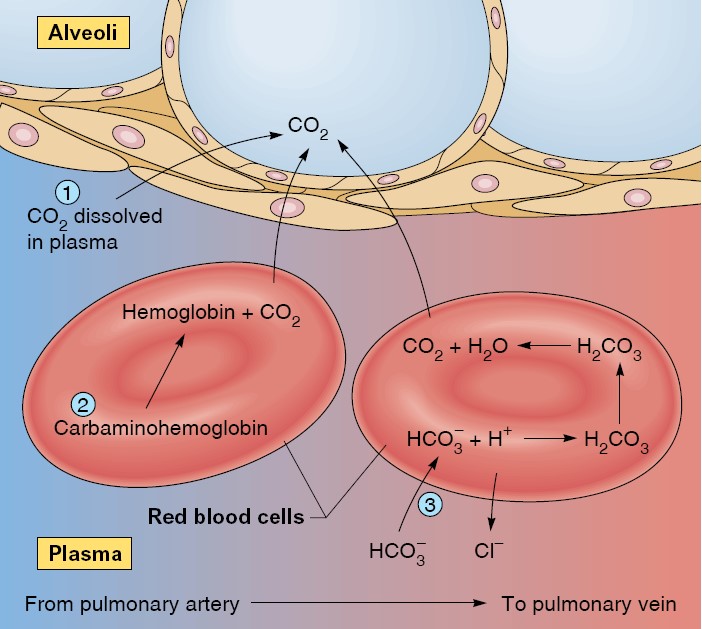
The Reverse Chloride Shift
When blood reaches the pulmonary capillaries, deoxyhemoglobin is converted to
oxyhemoglobin. Because oxyhemoglobin has a weaker affinity for H+ than does
deoxyhemoglobin, hydrogen ions are released within the red blood cells. This
attracts HCO3− from the plasma, which combines with H+ to form carbonic acid: H+
+ HCO3− → H2CO3. Under conditions of lower PCO2, as occurs in the pulmonary
capillaries, carbonic anhydrase catalyzes the conversion of carbonic acid to
carbon dioxide and water:
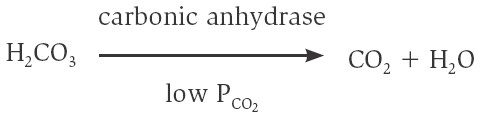
In summary, the carbon dioxide produced by the cells is converted within the
systemic capillaries, mostly through the action of carbonic anhydrase in the red
blood cells, to carbonic acid. With the buildup of carbonic acid concentrations
in the red blood cells, the carbonic acid dissociates into bicarbonate and H+,
which results in the chloride shift. A reverse chloride shift operates in
the pulmonary capillaries to convert carbonic acid to H2O and CO2 gas, which is
eliminated in the expired breath. The PCO2, carbonic acid, H+, and bicarbonate
concentrations in the systemic arteries are thus maintained relatively constant
by normal ventilation. This is required to maintain the acid-base balance of the
blood.
Pulmonary Volumes
The significance of each of pulmonary volumes is the following:
1. The tidal volume is the volume of air inspired or expired with each
normal breath; it amounts to about 500 milliliters in the average adult male.
2. The inspiratory reserve volume is the extra volume of air that can be
inspired over and above the normal tidal volume when the person inspires with
full force; it is usually equal to about 3000 milliliters.
3. The expiratory reserve volume is the maximum extra volume of air that
can be expired by forceful expiration after the end of a normal tidal
expiration; this volume normally amounts to about 1100 milliliters.
4. The residual volume is the volume of air remaining in the lungs after
the most forceful expiration; this volume averages about 1200 milliliters.
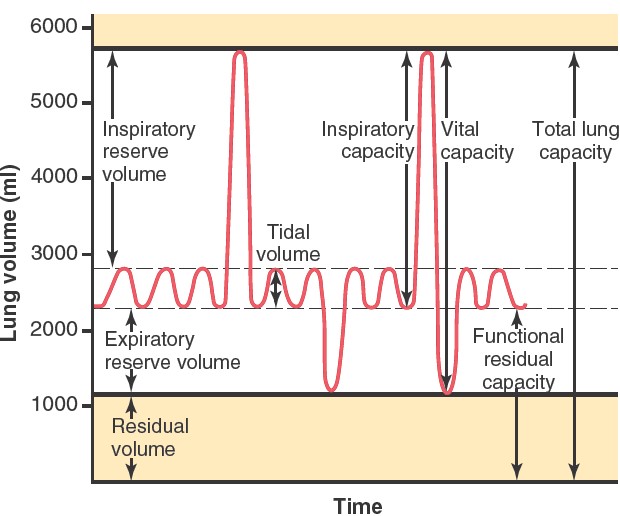
Pulmonary Capacities
In describing events in the pulmonary cycle, it is sometimes desirable to
consider two or more of the volumes together. Such combinations are called
pulmonary capacities. The important pulmonary capacities, which can be
described as follows:
1. The inspiratory capacity equals the tidal volume plus the
inspiratory reserve volume. This capacity is the amount of air (about 3500
milliliters) a person can breathe in, beginning at the normal expiratory level
and distending the lungs to the maximum amount.
2. The functional residual capacity equals the expiratory reserve
volume plus the residual volume. This capacity is the amount of air
that remains in the lungs at the end of normal expiration (about 2300
milliliters).
3. The vital capacity equals the inspiratory reserve volume plus
the tidal volume plus the expiratory reserve volume. This capacity
is the maximum amount of air a person can expel from the lungs after first
filling the lungs to their maximum extent and then expiring to the maximum
extent (about 4600 milliliters).
4. The total lung capacity is the maximum volume to which the lungs can
be expanded with the greatest possible effort (about 5800 milliliters); it is
equal to the vital capacity plus the residual volume.
All pulmonary volumes and capacities are usually about 20 to 25 percent less in
women than in men, and they are greater in large and athletic people than in
small and asthenic people.
ABBREVIATIONS AND SYMBOLS USED IN PULMONARY FUNCTION STUDIES
Spirometry is only one of many measurement procedures that the pulmonary
physician uses daily. Many of these measurement procedures depend heavily on
mathematical computations. To simplify these calculations, as well as the
presentation of pulmonary function data, several abbreviations and symbols have
become standardized. Using these symbols, we present here a few simple algebraic
exercises showing some of the interrelations among the pulmonary volumes and
capacities; the student should think through and verify these interrelations.
VC = IRV + VT + ERV
VC = IC + ERV
TLC = VC + RV
TLC = IC + FRC
FRC = ERV + RV
|
Table :
Average Pulmonary Volumes and Capacities for a Healthy, Young
Adult Man |
|
|
Pulmonary Volumes and Capacities
|
Normal Values (ml)
|
|
Volumes
|
|
|
Tidal volume |
500
|
|
Inspiratory reserve volume |
3000
|
|
Expiratory volume |
1100
|
|
Residual volume |
1200
|
|
Capacities
|
|
|
Inspiratory capacity |
3500
|
|
Functional residual capacity |
2300
|
|
Vital capacity |
4600
|
|
Total lung capacity |
5800
|
During respiration, the volume and functioning of
lungs, represented by different volume and capacity. Volume represents a
particular state whereas capacity refers to the combination of different
volumes.
VARIOUS BUFFERS OF BODY FLUIDS
Body fluid contains four buffer system namely
a. Bicarbonate buffer system
b. Phosphate buffer system
c. Protein buffer system
d. Hemoglobin buffer system
a. Bicarbonate buffer system
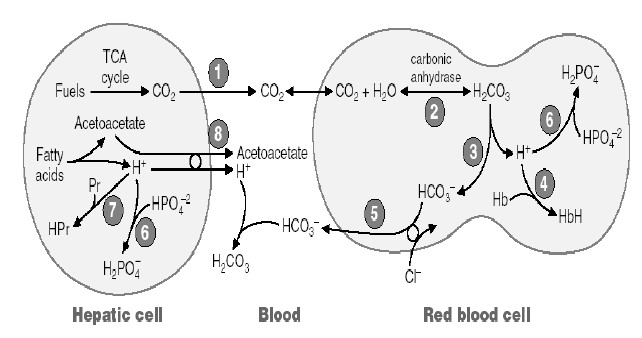
The carbon dioxide released during fuel metabolism reacts with water by the action of enzyme carbonic anhydrase to form carbonic acid [H2CO3]. Carbonic acid is a weak acid that partially dissociated into bicarbonate ion [HCO-3] and H+ ion.
CO2 + H2O à H2CO3 à H+ + HCO-3
As base is added and H+ removed, carbonic acid dissociates into hydrogen ion and bicarbonate ions, and dissolved CO2 reacts with water to replenish the carbonic acid levels. When CO2 levels increased, it forms more amount of carbonic acid which inturn dissociated into hydrogen ion and bicarbonate ions. Thus bicarbonate buffer functions as buffer system.
b. Phosphate buffer system
Phosphoric acid dissociates into H+ ions and dihydrogen phosphate [H2PO4-] ions with pKa of 2.15. Dihydrogen phosphate [H2PO4-] ion dissociates into H+ ions and Monohydrogen phosphate [HPO42-] ions with pKa of 7.2 whereas Monohydrogen phosphate ions dissociates into hydrogen ion and phosphate PO43- anions with pKa of 12.4. From the dissociation constant values, it was clearly understood that phosphate act as effective buffer in blood (pH = 7.4).
H3PO4 à H+ + H2PO4- à H+ + HPO42- à H+ + PO43-
But, phosphate concentration is very low in blood, thus, phosphate buffer, plays major role as a intracellular bufer in red blood cell and other types of cells where their concentration higher than blood and interstitial fluid. The sodium salts of phoshoric acid also act as buffer system.
c. Protein buffer system
Plasma proteins actually responsible for protein buffer system. The buffering capacity of proteins depends upon the pK inonizable group of aminoacid side chains. Histidine amino acid plays vital role as buffering agent because its imidazole group pK value is 6.7 and it is the more effective contributor for protein buffer system. The plasma proteins responsible for the 2% buffering capacity of plasma.
Protein-H à Protein- + H+
d. Hemoglobin buffer systems
Hemoglobin present in erythrocytes also plays important role as buffering agent. It mainly buffers the acids produced during gaseous transport between lungs and tissues.
HHb à Hb- + H+
At tissue levels, H+ ions released from carbonic acid binds with hemoglobin and helps in the transport of CO2 as HCO3. In lungs, as hemoglobin combines with oxygen, it releases H+ ions, which inturn binds with HCO3- to form carbonic acid. Carbonic acid then dissociates into CO2 and water. Then, CO2 is exhaled.
Maintenance of pH
The body pH is maintained by buffer system along with the functions of Kidney and Lungs. The role of kidney and lungs in the maintenance of pH is as follows.
Role of Kidney
The kidney maintains pH by excreting either acidic or basic urine. Excretion of
acidic urine increases pH whereas basic urine excretion decreases pH in
extracellular. Large amount of H+
ions are secreted into the tubular lumen by tubular epithelial cells. If they
are removed in urine, it will increase pH of extracellular fluid. Large amount
of bicarbonate ions are also continuously secreted in the renal tubules, if they
are excreted in urine, then it reduces pH by retaining H+ ions.
If more hydrogen ions removed than bicarbonate ions then there will be a net
loss of acid whereas if more bicarbonate ions are filtered than hydrogen ions
are secreted, then there will be a net loss of base.
These functions achieved by mainly three components namely bicarbonate
buffer system, phosphate buffer system and ammonia. The role of these components
as follows:
Bicarbonate buffer system in kidney
The bicarbonate ions freely filtered through glomerulus, combines with hydrogen ions and forms carbonic acid which inturn dissociates into carbon dioxide and water. The carbon dioxide then diffused into tubular cells where it again combines with water to form carbonic acid in the presence of enzme carbonic anhydrase. Thus bicarbonate ions are reabsorbed. This pattern of H+ ion secretion occurs in proximal convoluted tube, ascending loop of henle and early part of distal convoluted tube.
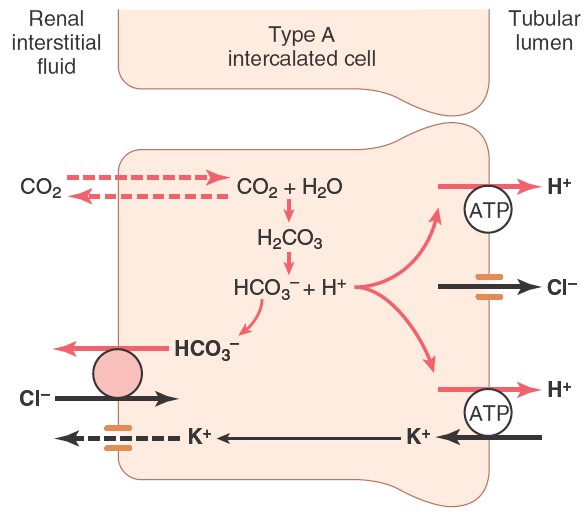
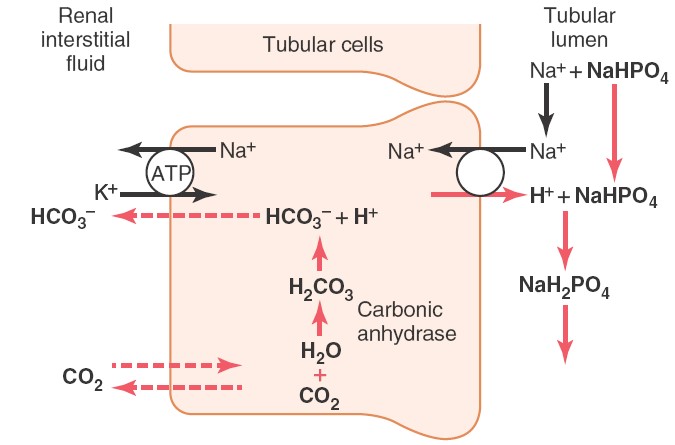
Phosphate buffer system in kidney
After the absorption of available bicarbonate ions in tubular filtrate,
remaining hydrogen ions interact with HPO2-4
or NaHPO-4 and forms H2PO-4or
NaH2PO4 which can be excreted in urine.
Thus, hydrogen ions removed from extracellular fluid.
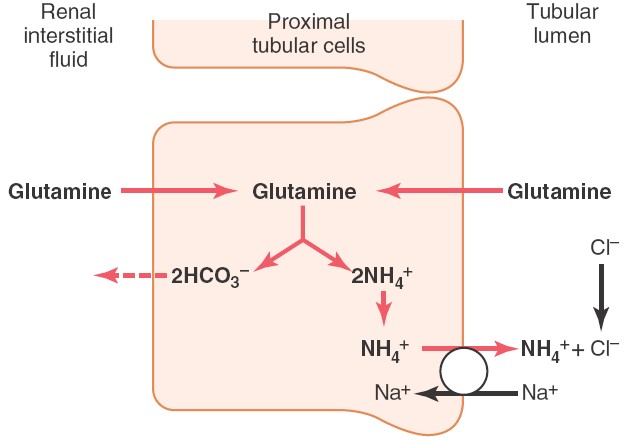
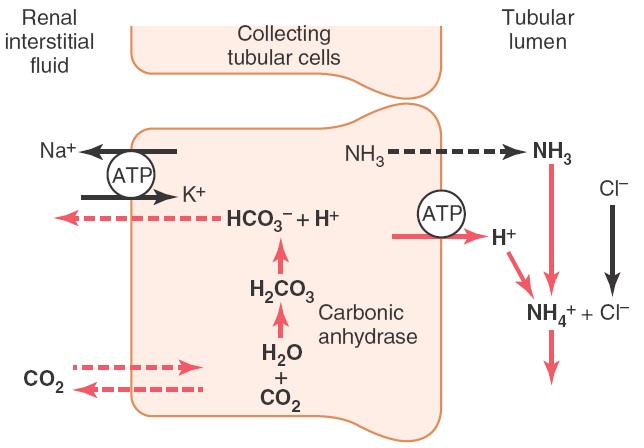
Ammonium buffer system in kidney
Ammonia produced from glutamine in tubular epithelial cells, can freely diffused
into tubular lumen. This ammonia
then combines with hydrogen ions to form ammonium ions which can be easily
excreted in urine. Thus, hydrogen
ions are removed from extracellular fluid.
Role of Lung
The second line of defense against maintenance of pH is the control of
extracellular concentrations of CO2 by the lungs. An increase in
ventilation removes CO2 from extracellular fluid which inturn reduces
hydrogen ion concentration. Conversely, decreased ventilation increases CO2,
thus increasing hydrogen ion concentration in the extracellular fluid.
Both bicarbonate buffer system and hemoglobin buffer system of
erythrocytes are important for the maintenance of pH by lungs. In general, the
overall buffering capacity of respiratory system is one to two times add great
as than the other chemical buffer system comibined of extracellular fluid.
Buffering Power of the Respiratory System.
Respiratory regulation of acid-base balance is a physiological type of buffer system because it acts rapidly and keeps the H+ concentration from changing too much until the slowly responding kidneys can eliminate the imbalance. In general, the overall buffering power of the respiratory system is one to two times as great as the buffering power of all other chemical buffers in the extracellular fluid combined. That is, one to two times as much acid or base can normally be buffered by this mechanism as by the chemical buffers.
Acid Base Disorders
Defective acid-base balance leads to acid-base disorders which are of mainly two types namely acidosis and alkalosis. These disorders arised mainly due to the alterations in bicarbonate ions and carbonic acid levels.
Acidosis
Increased level of carbonic acid or decreased level of bicarbonate ion results in acidosis. It will be of two types namely respiratory acidosis and metabolic acidosis.
Respiratory acidosis
Role of lungs in the elevation of carbonic acid is referred as respiratory
acidosis. The rise in arterial PCO2
due to decreased ventilation causes respiratory acidosis. The CO2
retained is in equilibrium with carbonic acid which inturn equilibrium with
bicarbonate and hydrogen ion. Thus
hydrogen ion concentration increased and pH decreased in extracellular fluid.
This inturn, promotes secretion of hydrogen ions in tubular lumen.
Metabolic acidosis
Metabolic abnormalities results in acidosis called as metabolic acidosis. When acids stronger than HHb and the other buffer acids are added to blood, metabolic acidosis is produced and when the H+ level falls as a result of addition of alkali or removal of acid, metabolic acidosis results. The hydrogen and bicarbonate ions combined to form carbonic acid. The carbonic acid converted into carbon dioxide and water then carbondioxie is excreted via lungs. In metabolic acidosis, an excess of hydrogen ion over bicarbonate ions occurs in tubular fluid primarily due to decreased filtration of bicarbonate ions. This decreased filtrations of bicarbonate ions is caused mainly by a decrease in extracellular concentrations of bicarbonate ions. The primary compensations for metabolic acidosis are increased ventilations and increased reabsorption of bicarbonate ions.
Alkalosis
Decreased level of carbonic acid or increased level of bicarbonate ion results in alkalosis. It will be of two types namely respiratory alkalosis and metabolic alkalosis.
Respiratory alkaosis
Role of lungs in the decrease of carbonic acid is refferred as respiratory alkalosis. The decrease in arterial PCO2 due to hyperventilation causes respiratory alkalosis. The CO2 removed is in equilibrium with carbonic acid which inturn equilibrium with bicarbonate and hydrogen ion. Thus hydrogen ion concentration decreased and pH increased in extracellular fluid. This inturn, decreases secretion of hydrogen ions in tubular lumen.
Metabolic alkalosis
Metabolic abnormalities result in alkalosis called as metabolic alkalosis. In metabolic alkalosis, plasma bicarbonate ions and pH increased. This is partly compensated by reduction in respiration rate which increases PCO2 and helps to return the extracellular pH toward normal. In addition,the increase in bicarbonate concentration in the extracellular fluid leads to the increase in filtered load of bicarbonate ions in tubular fluid over hydrogen ions. The excess bicarbonate ions excreted in urine without reabsorption due to lack of hydrogen ions. Thus by decreased ventilation and increased bicarbonate ions excretions, the raise in bicarbonate ions compensated.
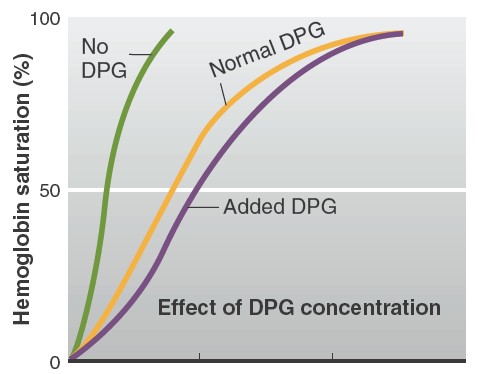
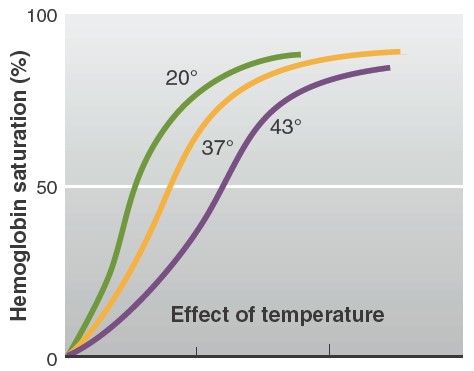
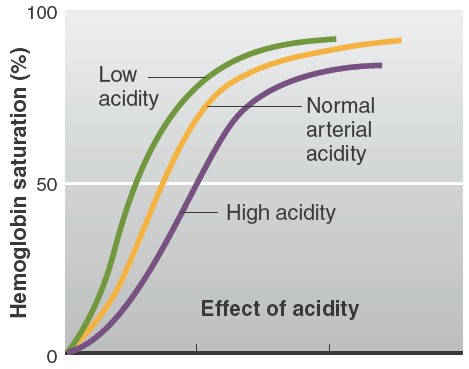
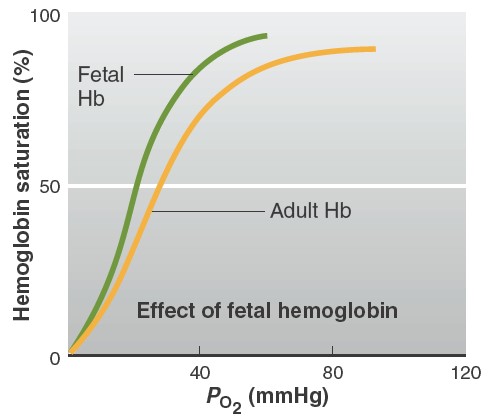
Factors affecting affinity of hemoglobin to oxygen
At any given PO2, other factors influence the degree of hemoglobin
saturation. These include blood PCO2, H1 concentration, temperature, the
concentration of a substance produced by erythrocytes called
2,3-diphosphoglycerate (DPG) (also known as bisphosphoglycerate
[BPG]), and the presence of a special kind of hemoglobin usually only found in
the fetal blood. An increase in DPG concentration, temperature, and acidity
causes the dissociation curve to shift to the right. This means that at any
given PO2, hemoglobin has less affinity for oxygen. In contrast, a
decrease in DPG concentration, temperature, or acidity causes the dissociation
curve to shift to the left, such that at any given PO2, hemoglobin has a
greater affinity for oxygen. The effects of increased PCO2, H+
concentration, and temperature are continuously exerted on the blood in tissue
capillaries, because each of these factors is greater in tissue capillary blood
than in arterial blood. The PCO2 is increased because of the carbon
dioxide entering the blood from the tissues. For reasons to be described later,
the H+ concentration is increased because of the increased PCO2
and the release of metabolically produced acids such as lactic acid. The
temperature is increased because of the heat produced
DPG, which is produced during glycolysis, reversibly binds with hemoglobin,
allosterically causing it to have a lower affinity for oxygen (see Figure
13.29). Erythrocytes have no mitochondria
and, therefore, rely exclusively on glycolysis. Consequently, erythrocytes
contain large quantities of DPG, which is present in only trace amounts in cells
with mitochondria. The net result is that whenever DPG concentrations increase,
there is enhanced
Effect of Carbon Monoxide on Oxygen Binding to Hemoglobin
Carbon monoxide
is a colorless, odorless gas that is a product of the incomplete combustion of
hydrocarbons, such as gasoline. It is a common cause of sickness and death due
to poisoning, both intentional and accidental. Its most striking
pathophysiological characteristic is its extremely high affinity—210 times that
of oxygen—for the oxygen-binding sites in
hemoglobin. For this reason, it reduces the amount of oxygen that combines with
hemoglobin in pulmonary capillaries by competing for these sites. It also exerts
a second deleterious effect: It alters the hemoglobin molecule shifting the
oxygen–hemoglobin dissociation curve to the left, thereby decreasing the
unloading of oxygen from hemoglobin in the tissues. The situation is worsened
because persons suffering from carbon monoxide poisoning do not show any reflex
increase in their ventilation.
REACTION
OF HEMOGLOBIN & OXYGEN
The dynamics of the reaction of hemoglobin with O2
make it a particularly suitable O2
carrier. Hemoglobin is a protein made up of four subunits, each of
which contains a heme moiety attached to a polypeptide chain. In normal
adults, most of the hemoglobin molecules contain two α and two β chains. Heme is
a porphyrin ring complex that includes one atom of ferrous iron. Each of the
four iron atoms in hemoglobin can reversibly bind one O2 molecule. The iron
stays in the ferrous state, so that the reaction is oxygenation (not
oxidation). It has been customary to write the reaction of hemoglobin with O2 as
Hb + O2
à HbO2. Because it contains four deoxyhemoglobin (Hb) units, the
hemoglobin molecule can also be represented as Hb4, and it actually reacts with
four molecules of O2 to form Hb4O8.
Hb4 + O2
à
Hb4O2
Hb4O2 + O2
à
Hb4O4
Hb4O4 + O2
à
Hb4O6
Hb4O6 + O2
à
Hb4O8
The reaction is rapid, requiring less than 0.01 s. The deoxygenation of Hb4O8 is
also very rapid. The quaternary
structure of hemoglobin determines its affinity for O2. In deoxyhemoglobin, the
globin units are tightly bound in a tense (T) configuration, which
reduces the affinity of the molecule for O2. When O2 is first bound, the bonds
holding the globin units are released, producing a relaxed (R) configuration,
which exposes more O2 binding sites. The net result is a 500-fold increase
in O2 affinity. In tissues, these reactions are reversed, resulting in O2
release. The transition from one state to another has been calculated to occur
about 108 times in the life of a red blood cell. The oxygen-hemoglobin
dissociation curve relates percentage saturation of the O2 carrying power
of hemoglobin (abbreviated as SaO2) to the Po2. This curve has a characteristic
sigmoid shape due to the T–R configuration interconversion. Combination of the
first heme in the Hb molecule with O2 increases the affinity of the second heme
for O2, and oxygenation of the second increases the affinity of the third, and
so on, so that the affinity of Hb for the fourth O2 molecule is many times that
for the first. Especially note that small changes at low Po2 lead to large
changes in SaO2.
