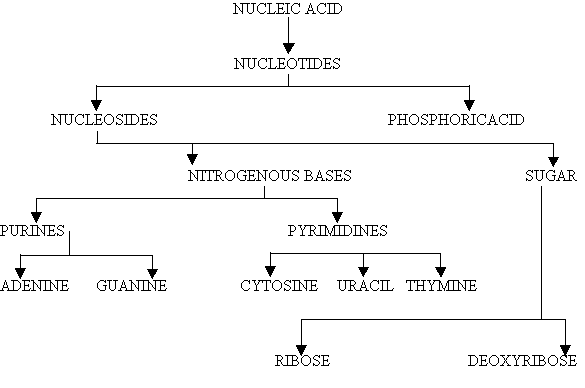
Nucleic acids are the principle genetic materials of all living organisms. They are complex macromolecule containing carbon, hydrogen, oxygen, nitrogen and phosphorous. Living organisms contain nucleic acid in the form of deoxyribonucleic acid (DNA) and ribonucleic acid(RNA). Some viruses contain only RNA(viroids) e.g. TMV(tobacco mosaic Virus) , poliomyelitis virus. Some viruses contain DNA only ex: bacteriophages, Vaccinia and Adenoviruses. In bacteria and higher cells both types of nucleic acids are found. The nucleic acids was first isolated in 1868 by miescher from a nuclei of pus cells on hospital bandages. He called it nuclei. He showed that the nuclein contained phosphorus, carbon, oxygen, hydrogen and nitrogen. Fischer (1880) identified that the Nucleic contains two types of nitrogenous bases and they are purines and pyrimidines. Altmann (1889) gave the term nucleic acid to replace nuclein. The name nucleic acid was given to it due too its acidic property. Levene identified a five-carbon sugar ribose in 1910. He later discovered that there was another type of nucleic acid, which contains deoxyribose. Nucleic acid that contains ribose sugar is called the ribonucleicacid (RNA) and that which contains deoxysugar named as the deoxyribonucleic acid. He also suggested that phosphorus is present in nucleic acid as a phosphate group. Upon hydrolysis, Nucleic acids yield 3 components namely phosphoric acid, a pentose sugar and nitrogenous bases.

PHOSPHORIC ACID:
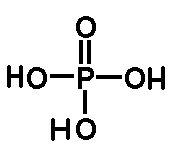
The molecular formula of phosphoric acid is H3PO4. It contains three monovalent hydroxyl groups and divalent oxygen atom, all linked toa pentavalent phosphorus atoms. Through 3’ -> 5’ phosphodiester bond, nucleotides linked in Nucleic acids.
PENTOSE SUGAR:
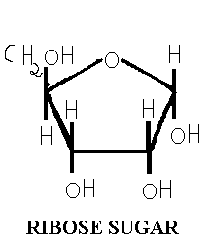
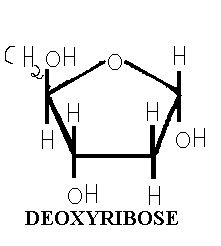
Five carbon sugars are called pentoses. RNA contains the pentose D-Ribose, while 2-deoxy D-ribose is found in DNA. In both cases, the pentose is in the five membered ring form known as furanose: D-ribofuranose for RNA and 2-deoxy D-ribose for DNA. When these ribofuranoses are found in nucleotides, their atoms numbered as 1’,2’,3’ and so on, to distinguish them from the ring atoms of the nitrogenous bases. An important property of the pentoses is their capacity to form esters with phosphoric acid. In this reaction, the OH- groups of the pentose, especially those at c3 and c5 are involved forming a 3’,5’-phosphodiester bond between adjacent pentose residues.
NITROGENOUS BASES:
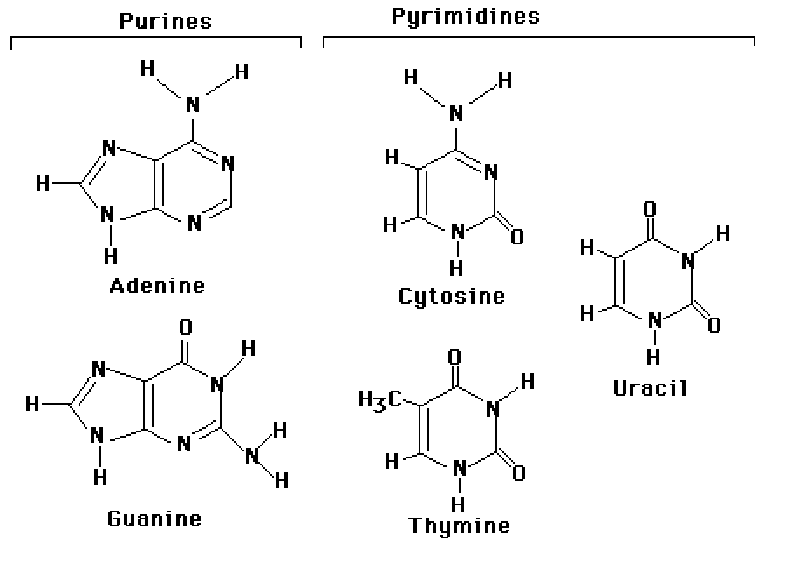
The bases of nucleotides and nucleic acids are derivatives of either pyrimidine or purine. Pyrimidines are sixmembered heterocyclic aromatic ring containing two nitrogen atoms. The common naturally occurring pyrimidines are cytosine,uracil and thymine. Cytosine and thymine found in DNA whereas cytosine and uracil are common in RNA. Various pyrimidine derivatives such as dihydrouracil are present as minor constituents in certain RNA molecules. The purine ring structure is represented by the ombination of pyrimidine ring with a five membered imidazole ring to yield a fused ring system. Adenine and guanine, the two common purines, are found in both DNA and RNA . Other naturally occurring purine derivatives include hypoxanthine , xanthine and uricacid. Hypoxanthine and xanthine are found only rarely as constituents of nucleicacids. Uric acid, the mose oxidized state for purine derivative, is never found in nucleic acids.
PROPERTIES OF BASES:
The pyrimidine ring system is planar, while the purine system deviates somewhat from planarity in having a slight pucker between its imidazole and ppyrimidine portions . Both are relatively soluble in water. The aromaticity of the pyrimidine and purine ring systems and the electron rich nature of their OH and NH2 groups made them with the capcity to undergo keto enol tautomeric shifts. Compounds that exist in two structural isomeric forms which are mutually interconvertable and exist in dynamic equilibrium are called tautomers and the phenomenon is termed tautomerism. Pyrimidines and purines exist as tautomeric pairs. The keto or imino tautomer is called a lactum whereas the enol or amino form is a lactim. It plays important role in hydrogen bond formation between bases.
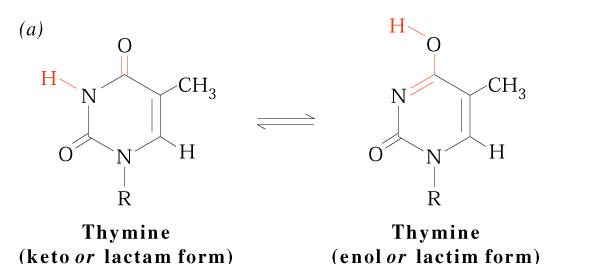
Another property of pyrimidines and purines is their strong absorbance of ultraviolet(UV) light, which is also a consequence of the aromaticity of their heterocyclic Ring structure.
NUCLEOSIDES:
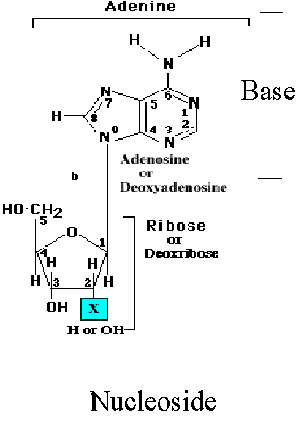
Nucleosides are compounds formed when a base is linked to a sugar via glycosidic bond. In nucleosides, the bond is an N-glycosidic because it connects the anomeric C’1 to N-1 of pyrimidine or to N-9 of a purine. The conformation of N-glycosidic bond is beta in purines and pyrimidines. Nucleosides are named by adding the ending –idine to the root name of a pyrimidine or –osine to the root name of purine.
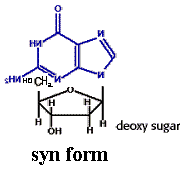
Nucleosides exist either of two conformations designated as syn and anti pyrimidine nucleosides favors anti conformation but purine nucleosides favors either syn or anti conformation. Nucleosides are much more water-soluble than free bases because of the hydrophilicity of the sugar moiety.
|
BASE |
|
SUGAR |
|
NUCLEOSIDES |
TRIVALNAME |
|
Adenine |
+ |
Ribose |
à |
Adenine ribonucleoside |
Adenosine |
|
Guanine |
+ |
Ribose |
à |
Guanine ribonucleoside |
Guanosine |
|
Cytosine |
+ |
Ribose |
à |
Cytosine ribonucleoside |
Cytidine |
|
Thymine |
+ |
Ribose |
à |
Thymine ribonucleoside |
Thymidine |
|
Uracil |
+ |
Ribose |
à |
Uracil ribonucleoside |
Uridine |
|
Adenine |
+ |
Deoxyribose |
à |
Adenine deoxy
ribonucleoside |
Deoxyadenosine |
|
Guanine |
+ |
Deoxyribose |
à |
Guanine deoxy
ribonucleoside |
Deoxyguanosine |
|
Cytosine |
+ |
Deoxyribose |
à |
Cytosine deoxy
ribonucleoside |
Deoxycytidine |
|
Thymine |
+ |
Deoxyribose |
à |
Thymine deoxy
ribonucleoside |
Deoxythymidine |
|
Uracil |
+ |
Deoxyribose |
à |
Uracil deoxy
ribonucleoside |
deoxyuridine |
SYN AND ANTI CONFORMATION OF NUCLEOSIDES:
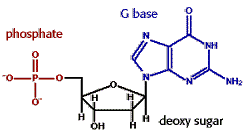
Mainly nucleosides serve as component parts of nucleotides. In exception, adenosine function as an autocoid or local hormone. Adenosine acts locally on specific cells to cause blood vessels dilation, smooth muscle contraction, neuronal discharge, neurotransmitter release and influence fat metabolism. Adenosine is also implicated in sleep regulation. Increased level of adenosine in brain cells promote sleepiness. Caffeine promote wakefulness by blocking the interaction of extracellular adenosine with its neuronal receptors.
Recently two nucleoside analogues 3’-azidodeoxythymidine and 2’,3’-dideoxycytidine have been therapeutically used for the treatment of AIDS(Acquired immune deficiency syndrome) patients. They inhibit DNA synthesis by HIV by causing premature chain termination due to its analogues nature to dTTP and dCTP.
NUCLEOTIDES:

Nucleotides result when phosphoric acid is esterified to a sugar hydroxyl group of a nucleoside. Ribose sugar bas three hydroxyl groups at C-2’, C-3’ and C-5’ where as deoxysugar has two hydroxyl groups at C-3’ and C-5’. The vast majority of monomeric nucleotides in the cell are ribonucleotides or deoxyribonucleoties having 5’-phosphate groups. Nucleotides have acidic properties because of this they are referred by names like adenylic acid, guanylic acid etc.,
|
NUCLEOSIDES |
|
PHOSPHORICACID |
|
NUCLEOTIDES |
TRIVAL NAME |
|
Adenosine |
+ |
Phosphoric acid |
à |
Adenosine
monophosphate(AMP) |
Adenylic acid |
|
Guanosine
|
+ |
Phosphoric acid |
à |
Guanosine
monophosphate(GMP) |
Guanylic acid |
|
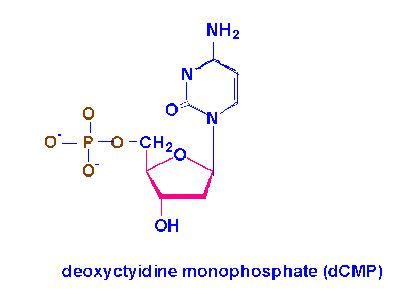
Cytidine |
+ |
Phosphoric acid |
à |
Cytidine monophosphate(CMP) |
Cytidylic acid |
|
Uridine |
+ |
Phosphoric acid |
à |
Uridine monophosphate(UMP) |
Uridylic acid |
|
Deoxyadenosine |
+ |
Phosphoric acid |
à |
deoxyadenosine
monophosphate(dAMP) |
Deoxy adenylicacid |
|
Deoxyguanosine |
+ |
Phosphoric acid |
à |
Deoxyguanosine
monophosphate(dGMP) |
Deoxyguanylic acid |
|
Deoxythymidine |
+ |
Phosphoric acid |
à |
Deoxythymidine
monophosphate(dTMP) |
Deoxythymidilic acid |
|
deoxycytidine |
+ |
Phosphoric acid |
à |
Deoxycytidine
monophosphate(dCMP) |
Deoxycytidylicacid |
Nucleoside monophosphates in which the phosphoric acid is esterified to two of the available ribose hydroxyl groupsare found in all cells. Formation of two ester linkages with one phosphate results in cyclic structure ex: cyclic CMP or cAMP and cyclicGMP or cGMP . they are act as secondary messenger which inturn act as important regulators of cellular metabolism. Additional phosphate groups can be linked to the phosphoryl group of a nucleotide through the formation of phosphoric anhydride linkages. Addition of second phosphate group to AMP creates adenosine-5’-diphosphate or ADP and adding a third yields adenosine-5’-triphosphate or ATP. The respective phosphate groups are designated by the Greek letters alpha , beta and gama starting with the alpha phosphate as the one linked directly to pentose.
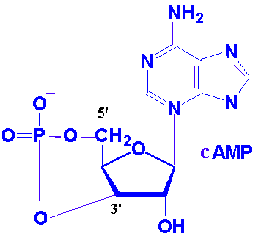
Nucleoside-5’-triphosphates are indispensable agents because they possess high energy which is required to do biological work. ATP has been termed the energy currency of the cell. GTP is the major energy soruce for protein synthesis. CTP is an essential metabolite in phospholipid synthesis and UTP forms activated intermediates with sugars that go on to serve as substrates in the biosynthesis of complex carbohydrates and polysaccharides. The four NTPs and dNTPs are the substrates for the synthesis of nucleicacids. Nucleotides serve as information symbols by transferring either phosphate or pyrophosphate group to substrates.
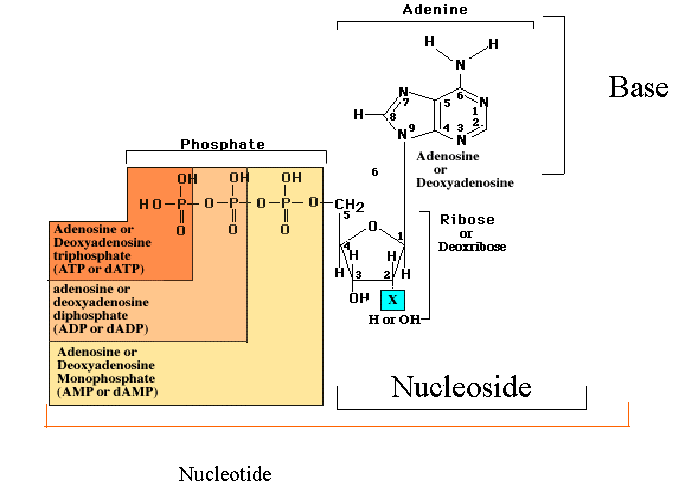
Adenosine nucleotide found to be important part of several coenzyme and cofactors. Example : NAD+ , FAD, Coenzyme A etc., they differ from eachother except for the presence of adenosine. When adenosine moiety removed from them actually reduces the activity of the enzyme. For example activity of the enzyme beta ketoacyl coA transferase of lipid metabolism reduced to 106 .
NUCLEIC ACIDS:
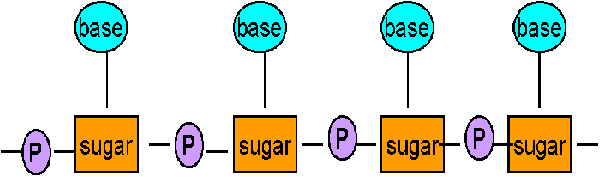
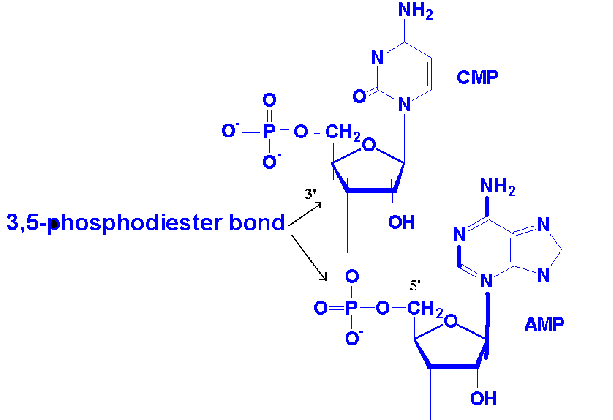
Nucleic acids are linear polymers of nucleotides linked 3’ to 5’ by phosphodiester bonds. They are formed as 5’ nucleosid monophosphates are successively added to the 3’ hydroxyl group of the preceeding nucleotide polymers of ribonucletides are named ribonucleic acid or RNA . Deoxy ribonucleotide polymers are called deoxyribonucleic acid or DNA. Shorthand notations for polynucleotide structures is as follows:
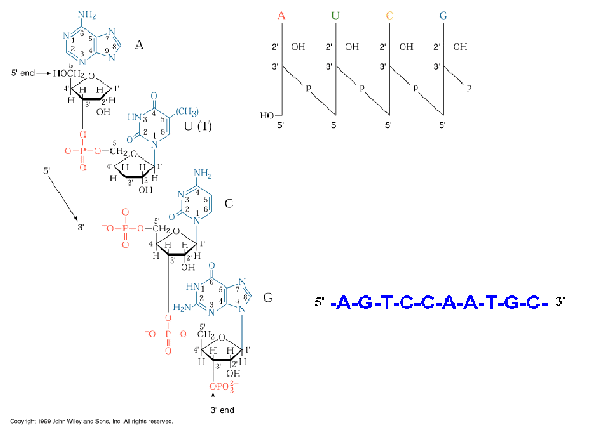
There are two major classes of nucleic acids. They are DNA and RNA. DNA has only one biological role i.e., it has the information to make all the functional macromolecules of the cell. DNA is chemically polydeoxy ribonucleotides. DNA consists of two polynucleotide strands wound together to form a long, slender, helicalmolecule, the DNA double helix. DNA has deoxyribose as pentose. It also consists thymine ,cytosine, guanine and adenine as bases.
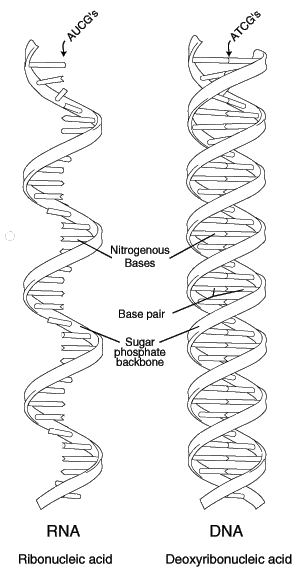
RNA occurs in multiple copies. Cells contain upto eigth times as much RNA as DNA. RNA has ribose as sugar. It also has uracil,cytosine, guanine and adenine as bases. RNA is chemically polyribonucleotides. RNA have major and minor classes. They are as follows :
|
RNA CLASS |
FUNCTION |
|
MAJOR CLASSES: |
|
|
mRNA (messenger RNA) |
The RNA transcribed
from protein encoding genes which carries the message for translation |
|
hnRNA (heterogenous
nuclearRNA) |
Prespliced mRNA. The
unmodified transcript of eukaryotic genes. |
|
tRNA (transfer or soluble
RNA) |
The adaptor molecule
which transfers amino acid from aminoacid pool to site of protein
synthesis(ribosomes) |
|
rRNA (ribosomal or
insolubleRNA) |
Major structural
component of ribosomes, required for protein synthesis. |
|
MINOT CLASSES: |
|
|
iRNA (initiator RNA) |
The shorter RNA
sequences used as primers for DNA Synthesis |
|
SnRNA (small
nuclearRNA) uRNA(uridine rich RNA) |
Low molecular weight
RNA molecules found in the nucleoplasm which facilitate the splicing of
introns and other processing reactions. |
|
SnoRNA(small nucleolar
RNA) |
Low molecular weight
RNA found in the nucleolus probably involved in the processing of rRNA |
|
ScRNA(small
cytoplasmic RNA) |
Low molecular weight
RNA molecules found in cytoplasm with various functions. Examples are 7s
RNA Which is part of the
signal recognition particle(SRP) and pRNA(prosomal RNA)
a small RNA associated with approximately 20 proteins and found
packaged with mRNA in the mRNP or
inforsome which may have a global regulatory effect on gene expression. |
|
Telomerase RNA |
A nuclear RNA which
contains the template for telomere repeats and forms part of the enzyme
telomerase |
|
gRNA(guide RNA) |
An RNA which provides
the templat for RNA editing ex: B-48 and B-100 apoprotein formation. |
|
Antisense RNA or micRNA(mRNA
interfering complementary RNA) |
Antisense RNA is
complementary to mRNA and can
form a duplex with it to block protein synthesis. They are used in
regulation of protein synthesis, gene therapy etc., |
|
Ribozymes(RNA enzymes) |
RNA molecules which
can catalyze chemical reactions ex: ribonuclease P which can catalyze tRNA
processing. |
|
|
|
ADVANTAGEOUS NATURE OF DNA THAN RNA:
Two fundamental chemical differences distinguish DNA from RNA. They may I) DNA contains 2’-deoxy ribose instead of ribose
II) DNA contains thymine instead of uracil.
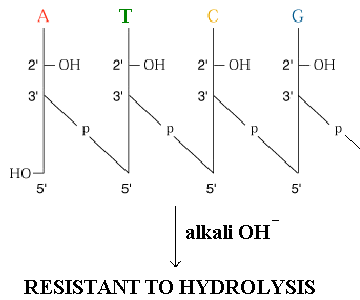
Absence of 2’-OH group mkes DNA resistance to alkaline hydrolysis. RNA susceptible to alkali hydrolysis because its vicinal 2’-OH group makes the 3’-phosphodiester bond susceptible too nucleophilic cleavage.
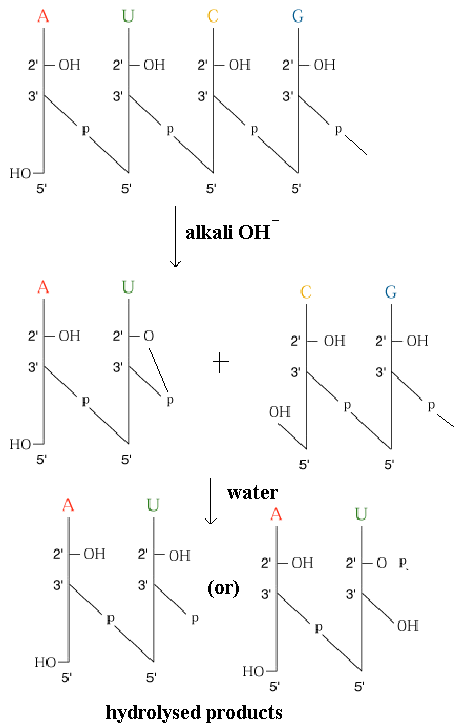
DNA consists thymine instead of uracil. The key observation is that cytosine deaminates to form uracil at a finite rate invivo. Because “C” in one DNA strand pairs with “G” in other strand, whereas “U” would pair with “A”, conversion of a “C” t o a “U” could potentially result in aheritable change of a CG pair to a UA pair.
Such a change in nucleotide sequence would constitute a mutation in the DNA. To prevent this reaction from leading too changes in nucleotide sequence, a cellular repair mechanisms “proof reads” DNA, and when a “U” araising from “C” deamination is encountered , it is created as inappropriate and is replaced by a “C” . If DNA normally contained “U” rather than “T”, this repair system could not readily distinguish “U” formed by “C” deamination from “U” correctly paired with “A”. Because of these two reasons , DNA is selectively advantageous for the heritable form of genetic information to be rather than RNA.
GENE ORGANISATION:
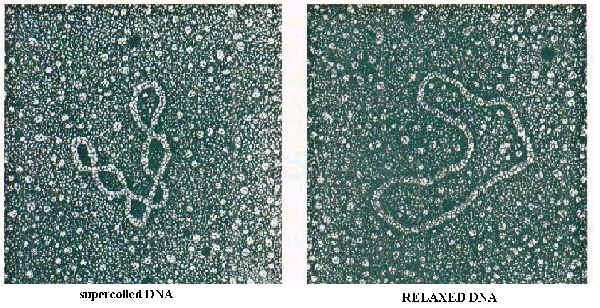
In prokaryotes, ex: E.Coli , a chromosome is a circular DNA molecule that is extensively looped and coiled. It consists of supercoiled DNA that is complexed with an RNA-protein core. In addition, the polyamines(ex: spermine and spermidine) may also assist in attaining the chromosome’s highly compressed structure.
3H+N-CH2-CH2-CH2-CH2-N+H2-CH2-CH2-CH2-NH3+ spermidine
3H+N-CH2-CH2-CH2-N+H2-CH2-CH2-CH2-CH2-N+H2-CH2-CH2-CH2-NH2+ spermine
In comparison to prokaryotes, the eukaryotes possess genomes which are extraordinary complex. Depending on species, the chromosomes of eukaryotes vary in both length and number. For example, human possess 23 pairs of chromosomes with a total of approximately three billion bps. The fruit fly Drosophila melanogaster has four chromosome pairs with 180 million bp, while corn(zea mays) has ten chromosome pairs with a total of 6.6million ba. In addition, each eukaryotic chromosome possess two unique structural elements centromeres and telomeres. A centromere is a specific AT rich DNA sequences associated with nonhistone proteins to form the kinetochore, which interacts with the spindle fibers during cell division. Telomeres are CCCA repetitive regions of DNA at the ends of the chromosome that prevent loss of coding sequences during DNA replication.
Each eukaryotic chromosome consists of a single linear DNA molecule complexed with histones to form nucleosome. Small amount of non-histone proteins also attached to DNA. There are five major classes of his tones name H1, H2A, H2B, H3 and H4. Octomeric core made up of two copies each of H2A, H2B, H3, and H4. Octomeric core made of two copies each of H2A, H2B, H3, and H4. H1 also binds to DNA separately.
The nucleosomes are coiled into a higher order of structure referred to as the 30nm fiber, which is referred, are solenoid. The 30nm fiber is further coiled to form 200nm filament (loops). The loops intern attached to a central core protein complex referred to as scaffold. This coil forms 1micrometere coil which intern forms chromatid of chromosome.
ISOLATION OF NUCLEIC ACIDS:
Nucleic acids’ isolation consists of four stages. They are
1.IDENTIFICATION
2.SEPARATION
3.
estimation
1.IDENTIFICATION:
Three main methods may be employed to determine or identify the localization of nucleic acid in individual cells or tissue sections.
A. Standing by one method known to be specific for either DNA or RNA
Example: Fuelgen which specific for DNA.usage of fuelgen depends upon the fact that the products of partial hydrolysis of DNA will restore the color of basic
Fuchsine, which has been decolorized with sulphuric acid. It is usually applied to tissue section in which DNA is partially hydrolyzed by being immersed in 1N HCL at 50-60* C for 10 min. The products of partial hydrolysis remain at the site of production. These products retain the color of fuchsine.
B. The location of nucleicacids can be determined by ultraviolet photomicrography. This method is based on the fact that no other protoplasmic conostituents absorb uv light at 260nm.
C. Tissue section may be examined before and after treatment with enzymes such as ribonuclease and deoxyribonuclease. The enzyme DNase or RNase may be used in conjugation with basic dyes such as Unnapappenheim mixture(mixture of pyronine and methyl green ) when the tissue section is incubated with aqueous solution of the enzyme, basophilic granules in the cell cytoplasm took pyronine and the chromatin component stained by methyl green. So, it appeared green in color.
2. SEPARATION:
Separtation of nucleicacid achieved in three steps. They are
a. Cell disruption
b. Crude separation
c. Purification
a. Cell disruption:
The disruption of cell may be brought about by any of the following methods such as alternate thawing and freezing of cells, subject the cells to an osmotic shock, treatment of the cells with detergents like SDS or treatment of the cells with enzymes like lyzozyme, hyaluronidase etc.,. For the isolation of nucleic acids, the last two methods are preferred.
b. Crude Separation:
After the disruption of the cell, the suspension subjected to centrifugation and nucleic acid separated from associated protein. Using phenol or repeated treatment with chloroform – isoamylalcohol or chloroform – octanol mixture, achieves this. Protein may be removed by digestion with enzyme such as protease, which digest proteins to amino acids.
ISOPYCNIC DENSITY CENTRIFUGATION:
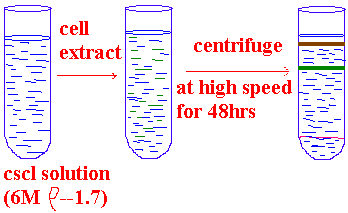
Cesium chloride centrifugation is an excellent an of removing RNA and proteins in the isolation of DNA. The density of DNA is typically slightly greater than 1.7g/cm3, while the density of RNA is more than 1.8g/cm3. Proteins have densities less than 1.3g/cm3. In cesium chloride solutions of appropriate density during centrifugation the DNA bonds present near the center of the tube, RNA pellets to the bottom and the proteins float near the top. Single stranded DNA is denser than double helical DNA.
In the case of DNA isolation done directly from the nuclear fraction which is obtained upon centrifugation. While in the case of RNA, certain modifications are employed depending upon the type of RNA to be isolated. Source for isolation varies for different type RNA ex: rRNA à ribosomal fraction, mRNA à polysomes, tRNA à cytoplasmic fraction.
FLOW CHART FOR DNA
ISOLATION:

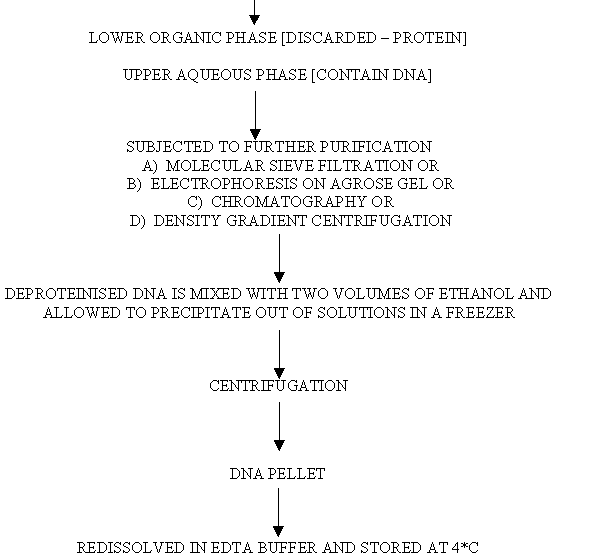
FLOW CHART FOR RNA ISOLATION:

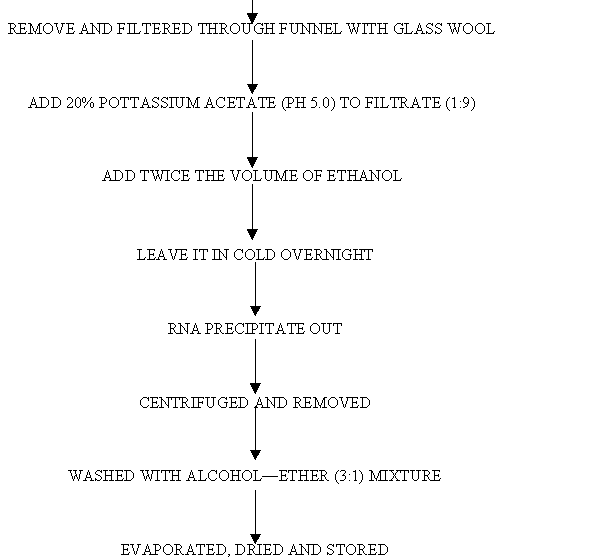
c. Purification:
The purification of nucleic acid is based on principles, which depend on differences in
a. Molecular size
b. Secondary structure
c. Base composition
a) Molecular Size:
Gel filtration on columns of dextran derivatives may be used for the
separation nucleic acid. Gel
filtration using play ethyl amide may also be used.
Cesium chloride density gradient also used for purification of DNA.
b) Secondary structure:
This is based on the differential affinity of single stranded and double
stranded nucleic acids to various materials.
Kieselguhr may be used for separation of single stranded DNA, which binds
very firmly to the columns of kieselguhr. Hydroxy appatite displays very strong affinity for double
stranded DNA column of the same may be used to separate dsDNA or DNA – RNA
Hybrid form single stranded DNA.
c) Base Composition:
3. ESTIMATION:
All chemical methods, which are devised, for the estimation of nucleic acid based on
a. Phosphorous
b. Purines or pyrimidines nitrogenous bases
c. Determination of total or relative ribose or deoxyribose sugar
In all the methods of estimation, finely divided tissues are extracted with acid usually trichloroaceticacid, perchloricacid followed by extraction with lipid solvent.
a) DETERMINATION OF PHOSPHOROUS
The extract obtained may be used for determination of phosphorous by Fiskee-Subbarow method in which blue colored complex is formed due to the reaction between inorganic phosphorous and molybdate. By using colorimeter, amount of phosphorous can be determined. Since, both DNA and RNA contains phosphorous this method used to estimate both DNA and RNA.
b) DETERMINATION OF NITROGENOUS BASES:
The nitrogenous bases of nucleic acid have characterized extinction co-efficient in UV region because of the presence of conjugated double bonds in their structure. Hence. The amount of nucleic acid in solution may be estimated by measuring extinction co-efficient in uvregion.
|
BASES |
ABSORBANCE AT
|
|
ADENINE |
260nm |
|
GUANINE |
250nm |
|
URACIL |
260nm |
|
THYMINE |
265nm |
|
CYTOSINE |
273nm |
Since both RNA and DNA consist of nitrogenous bases, this method is used to estimate both DNA and RNA.
c) DETERMINATION OF SUGARS:
When the amount of a particular nucleicacid (DNA or RNA) is to be estimated, the determination of the amount of sugar is ideal because deoxyribose sugar present in DNA and ribose sugar present in RNA. Thus, by estimating deoxyribose sugar and ribose sugar, estimation of DNA and RNA can be done respectively.
i) DETERMINATION OF DEOXYSUGAR:
The commonly employed method for estimation of deoxyribose is diphenylamine method. Deoxyribose in DNA, in presence of acid forms hydroxylevuline aldehyde which intern reacts with diphenylamine to give blue color. But only the deoxyribose of purine nucleotides reacts. Since, this method is specific for deoxysugar, this method used to estimate DNA.
ii) DETERMINATION OF RIBOSE SUGAR:
Acid hydrolysis of RNA releases ribose and this in presence of strong
acid, dehydrated to yield furfural. Orcinol
reacts with furfural in the presence of ferric chloride to give a green color.
Purine nucleotides are generally more reactive than pyrimidine
nucleotides. RNA only contains
ribose sugar, this method used to determine RNA only.