MAJOR HISTOCOMPATIBILITY COMPLEX (MHC)
The primary function of the immune system is the recognition and elimination of foreign cells and antigens that enter the body. Tissues and organs grafted from one individual to another member of the same species (allograft) are recognized as foreign and rejected. It was early work of Gorer in the 1930s on the antigens responsible for allograft rejection in inbred mice that led to the discovery of the major histocompatibility complex (MHC). Gorer identified two blood group antigen systems in mice, one of which antigen 1 was common to all the strains. Antigen 2 was found only in some strains and appeared to be responsible for allograft rejection. This was called the H-2 antigen where H for histocompatibility. By histocompatibility antigen is meant cell surface antigens that induce an immune response leading to rejection of allografts. The H-2 antigen system was found to be the major histocompatibility antigen for mice and coded for by a closely linked multiallelic cluster of genes, which was called the major histocompatibility complex. The development of congenic and recombinant strains of mice by snell enabled the detailed analysis of the various loci of this complex. Dausser pioneered studies on human leukocyte antigens, which were later found to be the major histocompatibility antigens in human beings. The genetic basis of immune response, which had been suggested by many early observations, was proved by Bnacerraf and colleagues, who established that the ability to respond immunologically to an antigen was conditioned by specific genes called the immune response (Ir) genes. For their work on MHC and the genetic control of immune response, Snell, Dausset and Benacerraf were awarded Nobel Prize for Medicine in 1980.
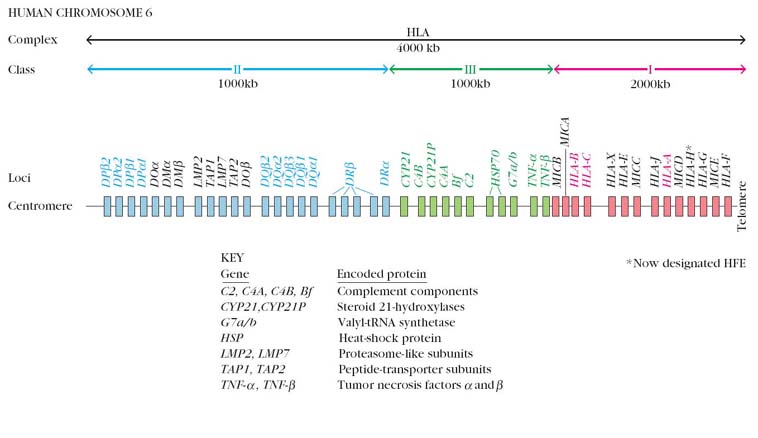
Early studies on MHC were carried out in mice. However, all species of animals examined subsequently were found to possess a similar complex of genes on a segment of one chromosome pair, coding for three different classes of proteins namely class –I proteins that determine histocompatibility and the acceptance or rejection of allografts, class-II proteins that regulate the immune response and class-III proteins, which are some components of the complement system. The name histocompatibility complex arose because its discovery was based on transplantation experiments and only later were the other two components of the complex identified. The major antigens determining histocompatibility in human beings are alloantigens, characteristically found on the surface of leucocytes. Human MHC antigens are therefore synonymous with human leukocyte antigen (HLA) and MHC complex of genes with the HLA complex.
MHC GENES:
Major histocompatibility complex refers to a cluster of genes responsible for immune response, transplantation antigens and proteins of the complement system etc. The MHC contains 3 classes of genes. Class I genes encode glycoprotein expressed on the surface of nearly all nucleated cells. The major function of class I products is the presentation of peptide antigens to cytotoxic cells. Class II MHC genes encode glycoproteins expressed primarily on antigen presenting cells. They present processed antigenic peptides to helper T cells. Class III MHC genes encode various secreted proteins that have immune functions including the synthesis of complements and molecules involved in inflammation.

MHC STRUCUTRE:
There were three group of MHC molecules namely MHC-I, MHC-II and class III molecules.
Structure of MHC-I molecules:
There are 2 subtypes of class I MHC molecules which are present on the cell surface. They are class Ia and class Ib molecules.
MIHC-Ia:
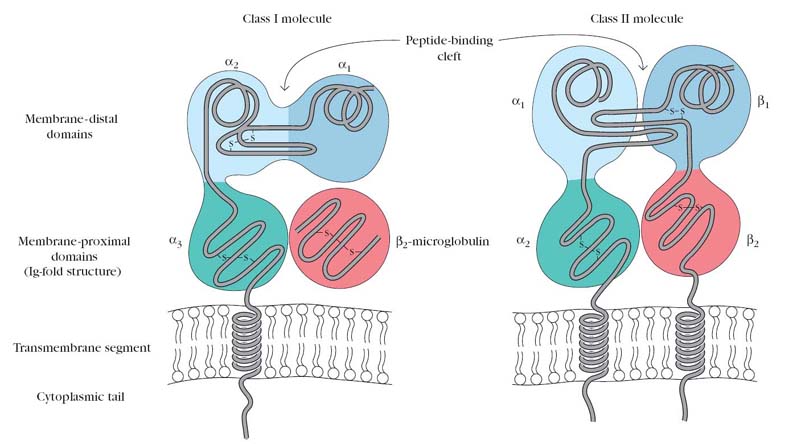
In humans there are 3 class Ia loci called A,B, and C which encode a single polypeptide chain. In human population as many as 23 a chain alleles can be found at the A locus and 9a chain alleles at B and 8a chain alleles at C. Class I MHC molecule have a transmembrane (hydrophobic) region and a cytoplasmic region (hydrophilic). There molecules contain a larger a chain associated noncovalently with much smaller B2 microglobulin. The a chain contain 3 extracellular domains a1, a2 and a3, each domain of which is about 100 amino acids log and contain a single interchain disulphide bond. In size and organization B2microglobin is similar to a3 domain. It does not, however, contain a transmembrane region and is noncovalently bound to class I glycoprotein. It is also coded by chromosome 15 and not by chromosome 6. Antigen binding cleft is contributed by a1 and a2 domain of a chain. The length of the peptide bound to MHC-Ia molecule is 8 – 10 amino acid residues. The anchoring residues usually present at the end of the peptide. Central region found to be elevated in its binding to MHC-Ia. The antigen presentation to MHC-Ia to TC cell is achieved because of the interaction between CD8+ and a3 domain of a chain of MHC-Ia.
MHC-Ib molecules:
The non polymorphic MHC class Ib molecules have a restricted tissue distribution. Some may be secreted and some are not encoded by genes located within MHC. In humans there are 6 class Ib loci HLA-E,F,G,H,J and X. HLA H,J and X are pseudogenes. HLA E and X are located between HLA-A and HLA-C and expressed on variety of cells. HLA F is differentially expressed in resting T-cells and skin cells. Due to the lack of polymorphism, Class Ib molecules can bind to only a limited range of peptides. They can act as receptor for specific, commonly encountered microbial antigens.
Structure MHC-II molecules:
The class-II molecules are found constitutively on the antigen presenting cells like B cells, dendritic cells and Macrophages etc., They may be induced as on T-cells, keratinocytes and vascular endothelial cells within the MHC class-II region of humans, five gene loci are arranged in the order of DP, DN, DO, DQ and DR. Within each locus, the genes for A chains are designated as A and the genes for B chains are designated as B. The DN locus contains only a singly A gene and do not contains a sing B gene, but the protein products of these genes have never been found. In contrast DP,DQ and DR loci are expressed. DP locus contains 2 each of A and B genes. The DQ locus contains two A and three B genes. In the DR locus only two B genes are present. Class II molecules show great polymorphism as exemplified by the fact that their amino acid sequence shows great variability.
MHC classII molecules are cell surface glycoproteins which contain 3 region namely, extracellular, transmembrane and cytoplasmic. It consists of tow non-covalently linked polypeptide chains called A and B. Each chain in class II molecules contains two external domains a1 and a2 in chain A and b1 and b2 in B chain. The membrane distal portion is composed of a1 and b1 domains and it is the antigen binding cleft for processed antigen. a2 and b2 domains forms the membrane proximal portion. Thus each chain consists of two extracellular domains, a connective peptide, transmembrane domain and a cytoplasmic domain. The presentation of exogenous antigen is the prime function of MHC classII molecules to TH cells. Due to the presentation of antigen to TH cells, both humoral and cell mediated immunity are activated. Because of this the antigen with the MHC class II molecules are called Immune associated antigen (Ia). The size of the peptide attached to MHC classII molecules varies from 12- 18 amino acid residues. The anchoring residues present throughout the peptide. CD4 of TH cells interact with the b2 domain of MHC classII molecule.
MHC CLASS III MOLECULES:
Several structurally and functionally diverse proteins are encoded with the third region of the MHC. These molecules include several complement components include C2, C4a, C4b and factor B, two steroid hydroxylase enzymes include 21-hydoroxylase A, 21-hydroxylase B, the inflammatory cytokines include tumor necrosis factor a and b (TNF a and b) and two heat shock proteins (HSP 70). Unlike classI and class II MHC molecules, the class III molecules are not present on the surface of membranes. They are not related structurally to class I and class II molecules and have no role in antigen presentations.
FUNCTIONS OF MHC:
Functions of MHC molecules can be studied under two headings namely immunological functions and non-immunological functions.
Immunological functions:
Under this category, transplantation, antigen processing and presentation, complement activation and anti-tumor activity might be studied.
Transplantation:
Before carrying out transplantation, HLA typing has to be carried out. Histocompatibility due to the presence of MHC plays crucial role for graft acceptance and graft rejection. Thus MHC was important for transplantation process.
Antigen Processing and Presentation:
Recognition of antigens by a T cell requires that peptides derived from the antigen be displayed within the cleft of an MHC molecule on the membrane of a cell. The formation of these peptide-MHC complexes requires that a protein antigen be degraded into peptides by a sequence of events called antigen processing. The degraded peptides then associate with MHC molecules within the cell interior, and the peptide-MHC complexes are transported to the membrane, where they are displayed which is known as antigen presentation.
Class I and class II MHC molecules associate with peptides that have been processed in different intracellular compartments. Class I MHC molecules bind peptides derived from endogenous antigens that have been processed within the cytoplasm of the cell (e.g., normal cellular proteins, tumor proteins, or viral and bacterial proteins produced within infected cells). Class II MHC molecules bind peptides derived from exogenous antigens that are internalized by phagocytosis or endocytosis and processed within the endocytic pathway.
The immune system uses two different pathways to eliminate intracellular and extracellular antigens. Endogenous antigens (those generated within the cell) are processed in the cytosolic pathway and presented on the membrane with class I MHC molecules; exogenous antigens (those taken up by endocytosis) are processed in the endocytic pathway and presented on the membrane with class II MHC molecules.
Cytosolic Pathway (Endogenous pathway):
In cytosolic pathway, endogenous antigens are thought to be degraded within the cytoplasm by the large LMP-containing proteasome complex. Peptides are then transported across the membrane of the RER by an ATP-binding transporter called TAP. The calnexin associated class I MHC molecule then associates with the transmembrane TAP protein and captures the peptides. Binding of the peptide to the calss I MHC molecule stabilizes the complex and causes its dissociation from calnexin. Finally, the class I MHC molecule-peptide complex is transported from the RER through the golgi complex to the plasma membrane.
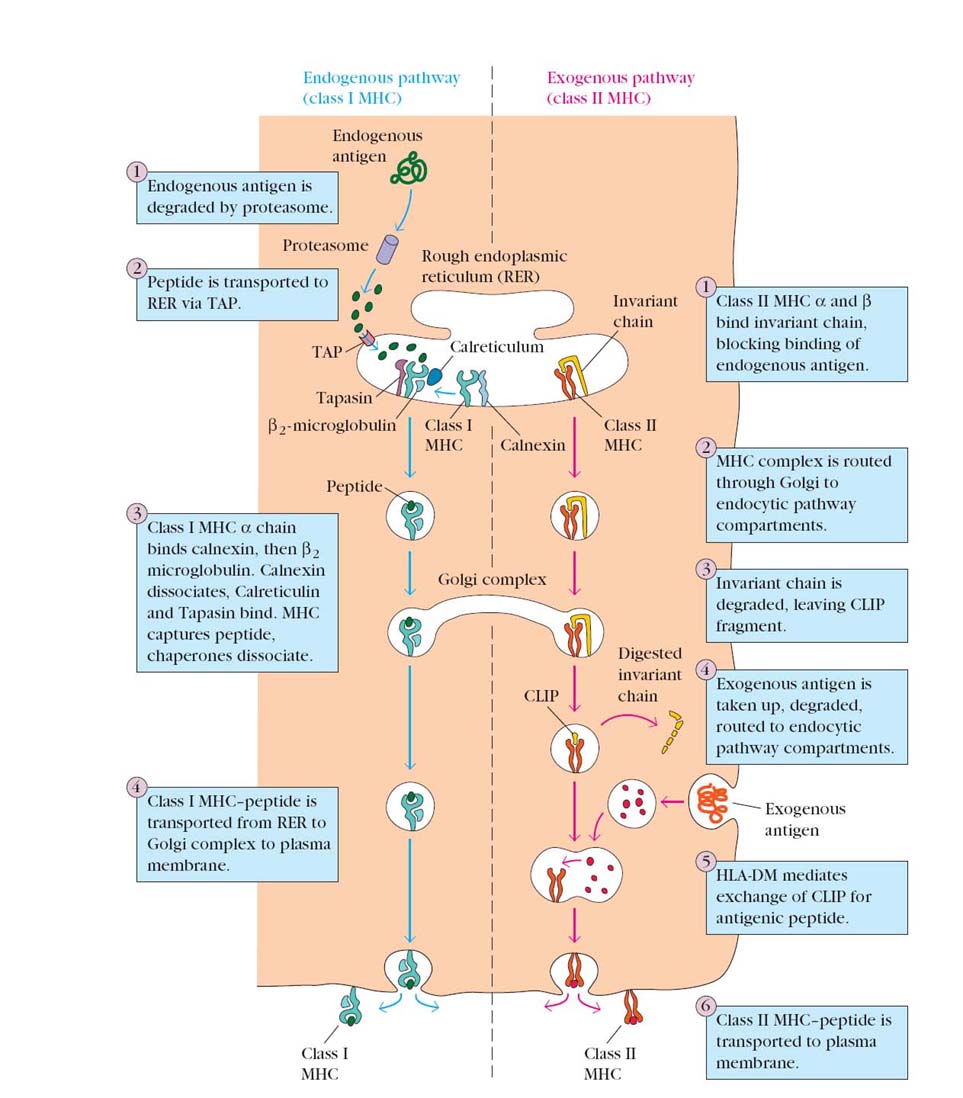
Endocytic Pathway (Exogenous pathway):
For Class II associated antigen presentation, extracellular proteins are internalized into endoscopes, where these proteins are proteolytically cleaved by enzymes that function at acidic pH. Newly synthesized class II MHC molecule associated with the Ii are transported from the ER to the endosomal vesicles. Here, the Ii is proteolytically cleaved and a small peptide remnant of the Ii, called CLIP, is removed from the peptide binding cleft of the MHC molecule by the HLA-DM molecules. The peptides that were generated from extracellular proteins then bin to the available cleft of other class II MHC molecule and the trimeric complex i.e class II MHC a and b chains and peptide, moves to and is displayed on the surface of the cell.
Complement activation:
Complements lime C2, C4 and Bf are produced from MHC genes. These complements required for all the three complements pathways. Thus, if there is any defect occurs in MHC region then it will affect complement system.
Anti-tumor Activity:
Tumor Necrosis factor plays important role anti-tumor activity. Since TNF was coded by MHC without which anti-tumor activity by TNF may not be possible.
Non-immunological functions:
Under this category, Steroidal metabolism and stress response plays vital role.
Steroidal Metabolism:
Steroid hydroxylase enzyme, 21-hydroxylase found to be an important regulatory enzyme of steroid metabolism. Defect in the action of this enzyme will affect the defective steroid metabolism which intern affects mineral metabolism and sex steroid defects.
Stress Response:
Heat shock proteins like HSP70 produced from MHC. HSP70 plays vital role in maintaining three dimensional protein structures during heat stress. This protein was gaining importance at present time due to the raise in the global temperature.
MHC RESTRICTION:
The importance of MHC antigen in immune reaction is indicated by the finding that T cells respond to antigens on the macrophages and other accessory cells only when they are presented along with the self-MHC antigen. This is known as MHC restriction. Both class I and class II antigens operate in this phenomenon. Cytotoxic T lymphocytes from immunized mice are able to kill and lyse virus infected target cells only when the T cells and target cells are of the same MHC type, so that the T cells can recognize the class I MHC antigens on the target cells. Helper T cells can accept antigen presented by macrophages only when the macrophages bear the same class II MHC molecules on the surface. For T cells participating in delayed type hypersensitivity the antigen has to be presented along with class II MHC determinants.
MHC AND IMMUNE RESPONSE:
There will be a good relationship present between MHC and Immune Response. Two explanations proposed to account for the variability in immune responsiveness observed among different haplotypes i.e. people with different MHC molecules.
Determinant selection model assumes that the structure of a particular MHC molecule determines the strength of its association with any given antigen. It is otherwise explained as in order to present an antigen, one should have specific type of MHC molecule. Absence of such MHC molecule will leads to immune unresponsiveness to a particular antigen.
Hole in the repertoire model proposed that an absence of T cells capable of recognizing a specific MHC-antigen molecule complex will leads to the immune unresponsive status. Thus in order to elicit immune response against a specific antigen both MHC and T-cells are important.
MHC AND DISEASE SUSCEPTIBILITY:
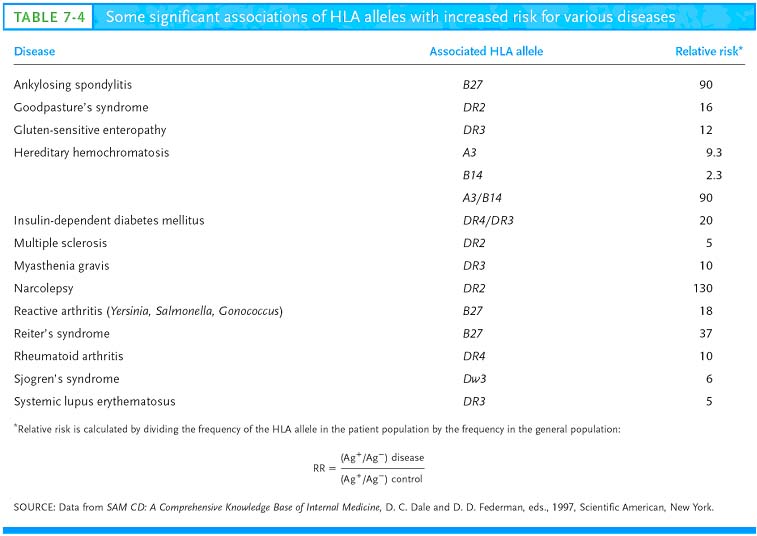
Some HLA alleles occur at a much higher frequency in those suffering from certain diseases than in the general population. The diseases associated with particular MHC alleles include autoimmune disorders, certain viral diseases, disorders of the complement system, some neurologic disorders, and several different allergies. The association between HLA alleles and a given disease may be quantified by determining the frequency of the HLA alleles expressed by individuals afflicted with the disease, then comparing these data with the frequency of the same alleles in the general population. Such a comparison allows calculation of relative risk. A relative risk value of 1 means that the HLA allele is expressed with the same frequency in the patient and general populations, indicating that the allele confers no increased risk for the disease. A relative risk value substantially above 1 indicates an association between the HLA allele and the disease. As Table 7-4 shows, individuals with the HLAB27 allele have a 90 times greater likelihood (relative risk of 90) of developing the autoimmune disease ankylosing spondylitis, an inflammatory disease of vertebral joints characterized by destruction of cartilage, than do individuals with a different HLA-B allele.
Case study report clearly indicates the relationship between MHC and diseases. For example, chickens expressing the MHC B19 allele are susceptible to the virus causing Marek’s disease, whereas birds expressing the B21 allele are not susceptible. An interesting link between the MHC and disease susceptibility in humans came from a study of Dutch immigrants to South America.
In 1845 a group of Dutch immigrants consisting of 367 individuals from 50 families emigrated from Europe to South America. Within two weeks of their arrival an epidemic of typhoid fever killed 50% of the immigrants and six years later an epidemic of yellow fever killed another 20% of the population. Thereafter the annual mortality rate was relatively low, and the survivors remained and intermarried. Recently the MHC polymorphism of the selected descendants of the survivors was analyzed and compared with that of a similar number of Dutch families living in the Netherlands. The descendants in South America exhibited significant relative decrease in some HLA alleles, such as B7 and significant increases in other alleles, such as B13, Bw38 and Bw50. It is hypothesized that the MHC alleles carried by the descendants enabled them to respond more effectively to pathogens endemic to their South American environment.