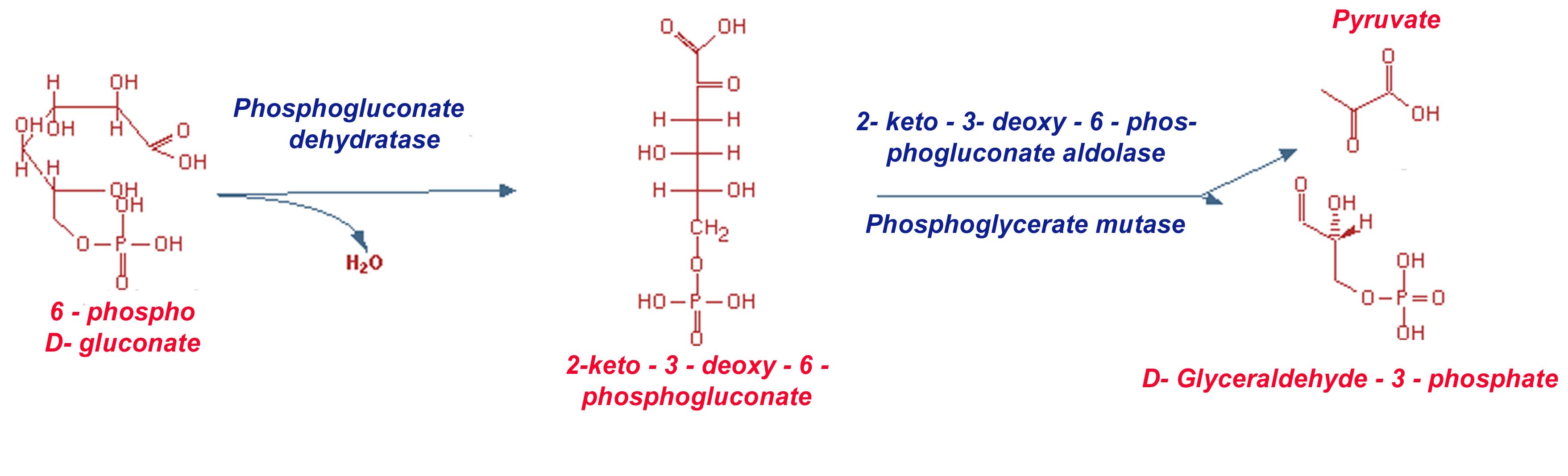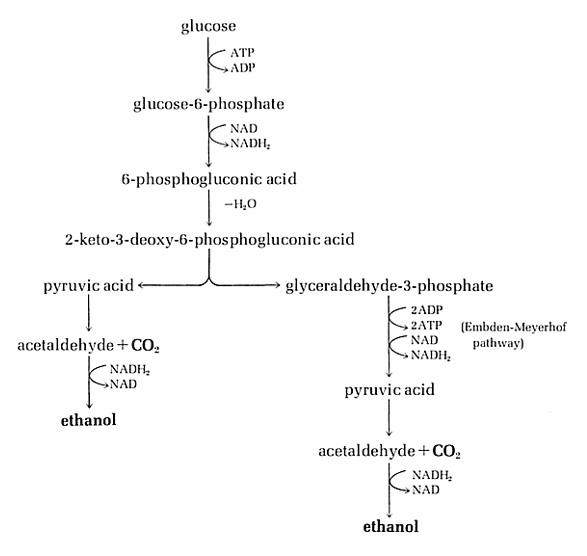
ENTNER-DOUDOROFF (ED) PATHWAY

The Entner-Doudoroff pathway joins the oxidative branch of the pentose phosphate pathway to glycolysis via two steps, catalyzed by inducible enzymes. The biochemical reactions of the Entner-Doudoroff pathway are a modification of the hexose monophosphate shunt, except that pentose sugars are not directly formed. The two pathways are identical up to the formation of 6-phosphogluconate and then diverge. In the Entner-Doudoroff pathway, no oxidative decarboxylation of 6-phosphogluconate occurs and no pentose compound is formed. For this pathway, a new 6 carbon compound intermediate (2-keto-3-deoxy6-phosphogluconate) is generated by the action of 6-phosphogluconate dehydratase (an Fe2+- and glutathione-stimulated enzyme); this intermediate compound is then directly cleaved into the triose (pyruvate) and a triose-phosphate compound (glyceraldehyde-3-phosphate) by the 2-keto-3-deoxy6-phosphogluconate aldolase. The glyceraldehyde-3-phosphate is further oxidized to another pyruvate molecule by the same enzyme systems that catalyze the terminal glycolytic pathway.
This pathway is important to E. coli because sugar acids, which are significant nutrients in both of its habitats, intestinal and aquatic, are metabolized via this pathway. Strains that are mutationally blocked in this pathway are unable to grow on gluconate, glucuronate, or galacturonate and are also unable to colonize the mouse intestine. The uronic acids feed into the pathway's sole intermediate, 2-keto-3-deoxy-6-phospho-gluconate so the dependence of their metabolism on an intact Entner-Doudoroff pathway is clear. The dependence of gluconate's metabolism on an intact Entner-Doudoroff pathway is less obvious because it is also metabolized via the pentose phosphate pathway. Enzymes of the Entner-Doudoroff pathway are induced by growth on gluconate, glucuronate, or methyl-β-D-glucuronide; phosphate or carbon limitation.